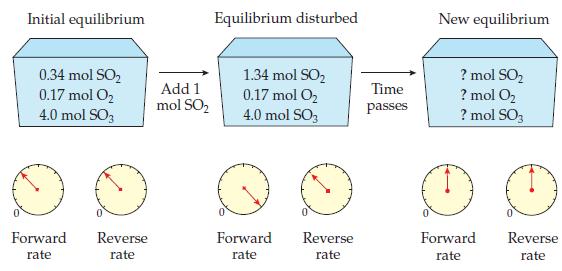
Which is the best choice for the amounts at the new equilibrium? (a) 0.34 mol SO 0.17
Question:
Which is the best choice for the amounts at the new equilibrium?
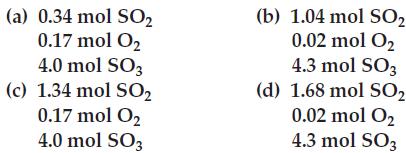
Transcribed Image Text:
(a) 0.34 mol SO₂ 0.17 mol O₂ 4.0 mol SO3 (c) 1.34 mol SO2 0.17 mol O₂ 4.0 mol SO3 (b) 1.04 mol SO₂ 0.02 mol O₂ 4.3 mol SO3 (d) 1.68 mol SO₂ 0.02 mol O₂ 4.3 mol SO3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
b Is the best choice Remember Le Chteliers princip...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
In WorkPatch 14.8, why are the rates of the forward and reverse reactions greater at the new equilibrium than at the initial equilibrium? Data from WorkPatch 14.8 Initial equilibrium 0.34 mol SO 0.17...
-
Which interface (FireWire, Thunderbolt, SCSI, iSCSI, InfiniBand, and Fibre Channel) is the best choice for a departmental server? Which type of multiplexing is most appropriate on a fiber optic...
-
The prisoners dilemma game is one of the most important models in all of social science: Most games of trust can be thought of as some kind of prisoners dilemma. Heres the classic game: Two men rob a...
-
Different theories about early childhood inform approaches to children's learning and development. Early childhood educators draw upon a range of perspectives in their work ..." (EYLF p.12)....
-
Determining Financial Statement Effects of Several Transactions For each of the following transactions of Pitt Inc. for the month of January 2012, indicate the accounts, amounts, and direction of the...
-
A survey of 1,085 adults asked "Do you enjoy shopping for clothing for yourself?" The results indicated that 51 % of the females enjoyed shopping for clothing for themselves as compared to 44% of the...
-
What are the totals of the debit and credit columns from the sales journal for the month of May? Debit amount: Credit amount:
-
The White Swan Talc Company purchased $120,000 of mining equipment for a small talc mine. The mining engineer's report indicates the mine contains 40,000 cubic meters of commercial quality talc. The...
-
If m f(x) M for a x b, where m is the absolute minimum and M is the absolute maximum of f on the interval [a, b], ther b m(b- a) f(x) dx M(b - a). Ja Use this property to estimate the value of the...
-
When writing an equilibrium constant expression for a chemical reaction: (a) It is important that the chemical equation be balanced. (b) The stoichiometric coefficients are used as exponents. (c)...
-
If we reverse the reaction for making NO, we have a reaction for the decomposition of NO: 2NO N 2 + O 2 (a) Write the K eq expression for this reaction and for the reaction to form NO(N 2 + O 2 ...
-
Indicate whether each of the following statements is true or false by writing T or F in the answer c olumn. An offer that includes specifi c time limits expires automatically when the time is up,...
-
Bims Corporation uses the weighted-average method in its process costing system. The Assembly Department started the month with 6,000 units in its beginning work in process inventory that were 90%...
-
For this activity you are going to research Canada's current (or as current as possible) Consumer Price Index (CPI) and also provide information about how the price of various items has changed over...
-
Design a database to automate the intake process for an estate planning attorney. The current process is based on capturing information in the attached form which every client completes....
-
A particle of mass m = 1 kg moves with an initial velocity of 1 m/s. It collides elastically with a second particle, of mass m = 2 kg, initially at rest. The angle between the initial trajectory and...
-
Four Seasons Company makes snow blowers. Materials are added at the beginning of the process and conversion costs are 60% completed. Other data for the September 2020 include: Beginning...
-
Jessica Brooks just slept through the class in which Professor Dominguez explained the concept of depreciation. Because the next test is scheduled for Wednesday, Jessica Brooks telephones Hanna...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
A float in an ocean harbor is made from a uniform foam having a specific weight of 12.00 lb/ft 3 . It is made in the shape of a rectangular solid 18.00 in square and 48.00 in long. A concrete...
-
Describe how the situation described in Problem 5.35 will change if the water level rises by 18 in during high tide. In Problem A float in an ocean harbor is made from a uniform foam having a...
-
A cube 6.00 in on a side is made from aluminum having a specific weight of 0.100 lb/in 3 . If the cube is suspended on a wire with half its volume in water and the other half in oil (sg = 0.85), what...
-
Dr. Minn is selling her physical therapy practice after owning the practice for 20 years. Dr. Linn is going to buy the radiology practice at a higher price because Dr. Minn has over 500 patients and...
-
Dr. Tier owns a non-profit agency in the local community that provides food, shelter, and counseling for disadvantaged youth and families. At the end of the year, the non-profit agency's difference...
-
Comparative financial statements for Weller Corporation, a merchandisi ompany, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total of...

Study smarter with the SolutionInn App


