(a) Compute e -a from Equation 8-44 for O 2 gas at standard conditions. (b) At what...
Question:
(a) Compute e-a from Equation 8-44 for O2 gas at standard conditions.
(b) At what temperature is e-a = 1 for O2?
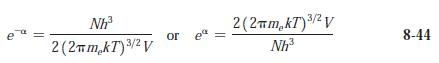
Transcribed Image Text:
e Nh³ 2 (2πm₂kT) ³/² V 3/2 or e 3/2 2(2πm₂kT) ³/² V Nh³ 8-44
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a b a 0 N V hc h 2 MkT 3 NA V...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The following is a list of 11 measurements. 64,-88, -81, 43, -14, 61, -99, 80, -24, -45, 12 Suppose that these 11 measurements are respectively labeled r, X, ..., X (Thus, 64 is labeled x, 88 is...
-
At what temperature does the rms speed of O2 molecules equal 400. m/s?
-
A tank contains 26.0kg of O2 gas at a gauge pressure of 8.70 atm. If the oxygen is replaced by helium, how many kilograms of the latter will be needed to produce a gauge pressure of 7.00 atm?
-
At what points are the function. y = x tan x 2 x + 1
-
Sketch the normal distribution with a. = 3 and = 3. b. = 1 and = 3. c. = 3 and = 1.
-
Graph the hyperbola. Give the domain, range, center, vertices, foci, and equations of the asymptotes. |(x+ 3)2 (y 2)2 16 = 1
-
Bierko Company is considering two different, mutually exclusive capital expenditure proposals. Project A will cost \($395,000\), has an expected useful life of 10 years, a salvage value of zero, and...
-
Dave and Reba Guerrera saved all their married life to open a bed and breakfast (B&B) named Tres Amigos. They invested $100,000 of their own money and the corporation issued common stock to them. The...
-
In 7-12, Identify all of the terms from the following list that apply to each figure: quadrilateral, parallelogram, rectangle, square, trapezoid, rhombus, kite. 7. 8. H
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Like 4 He, the most common form of neon, 20 Ne, is a rare gas and the 20 Ne atoms have zero spin and so are bosons. But unlike helium, neon does not become superfluid at low temperatures. Show that...
-
The molar mass of oxygen gas (O 2 ) is about 32 g/mol and that of hydrogen gas (H 2 ) about 2 g/mol. Compute (a) The rms speed of O 2 and (b) The rms speed of H 2 when the temperature is 0C.
-
How does the economists and accountants view of break-even charts differ?
-
What dual responsibility does a CPA have in relation to the concept of confidentiality?
-
During an audit of the personnel function, an auditor notes that there were several employee benefit programs and that participation in some of the programs is optional. Which of the following would...
-
Why may litigation with a client impair independence?
-
The first step in an operational audit is for the auditor to a. Define problem areas. b. Identify objectives of the company and the function being audited. c. Evaluate financial statements. d....
-
Under what conditions may members express unqualified opinions on financial statements that utilize accounting principles other than those issued by authoritative bodies?
-
Discuss the difference between profit maximization and shareholder wealth maximization. Which of these is a more comprehensive statement of a company's economic objectives?
-
Why is it necessary to study the diffusion of molecules in biological systems?
-
a. Write a realistic problem for which this is the correct equation. Be sure that the answer your problem requests is consistent with the equation given. b. Finish the solution of the problem. 60 N =...
-
A 500 g steel block rotates on a steel table while attached to a 2.0-m-long massless rod. Compressed air fed through the rod is ejected from a nozzle on the back of the block, exerting a thrust force...
-
A 500 g steel block rotates on a steel table while attached to a 2.0-m-long massless rod. Compressed air fed through the rod is ejected from a nozzle on the back of the block, exerting a thrust force...
-
Which of the following statements is correct? Select answer from the options below ASPE permits three measurement models for investments with no significant influence or control; IFRS permits only...
-
top inc acquired all of the outstanding stock of bottom co on january 1, 2020 for $276,000. Annual amortization of $21,000 restulted from this acquisition. Top reported net income of $80,000 in 2020...
-
The head of the accounting department of your company has determined that there is idle cash and is considering options for investing it. You are assigned to explain the costs and benefits of various...

Study smarter with the SolutionInn App


