Predict the outcome of the following reactions: CN CI 2 equiv. KNH lig. NH3, 33 C
Question:
Predict the outcome of the following reactions:
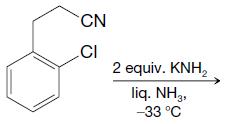
Transcribed Image Text:
CN CI 2 equiv. KNH₂ lig. NH3, 33 C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
For the first reaction the expected outcome is the formation of 1hexanol This is because the reactan...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Predict the outcome of the reactions represented by the following equations by using the activity series, and balance the equations. (a) Cu(s) + HCl(aq) (b) 12(s) + NaBr(aq)- (c) Mg(s) + CuSO (aq...
-
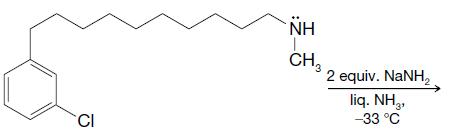
Predict the outcome of the following reactions: (a) (b) CN CI 2 equiv. KNH2 liq. NH3 -33 C NH CH3 2 equiv. NaNH liq. NH2 -33C Cl
-
Predict the outcome of the following reaction. -NH2
-
The Kc for the following reaction is 9.30 X 10^-2 at 25C:PCl5(g) <-> PCl3(g) + Cl2(g) How many moles & grams of PCl5 must be added to a 2-literflask to obtain a Cl2 concentration of 0.150M...
-
X 3 1 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Assume that two random variables (X, V) arc uniformly distributed on a circle with radius a. Then the joint probability density function is Find the expected value of X,fix- otherwise.
-
Comparative Data Comparative stockholders' equity sections from two successive years of balance sheets from Kirkwood, Inc., are as follows: No dividends were declared or paid during 2018. Required...
-
Beale Management has a noncontributory, defined benefit pension plan. On December 31, 2011 (the end of Beale's fiscal year), the following pension-related data were available: Required: 1. Prepare...
-
The partial autocorrelation function in time series analysis is useful for: Assessing the overall variability Identifying the seasonal component Estimating the trend component Determining the...
-
Blooming Flower Company was started in Year 1 when it acquired $60,000 cash from the issue of common stock. The following data summarize the companys first three years operating activities. Assume...
-
Provide a mechanism for the following reaction. NO NO 2 ss-sr NaOCH3 OH NO O=5 S=0 NO
-
Starting with aniline, outline a synthesis of each of the following: (a) p-Bromoaniline (b) o-Bromoaniline (c) 2-Bromo-4-nitroaniline (d) 4-Bromo-2-nitroaniline
-
Gandy Oil Incorporated has an account titled Oil and Gas Properties. Gandy paid $6,100,000 for oil reserves holding an estimated 300,000 barrels of oil. Assume the company paid $560,000 for...
-
How could you proceed to find out how many of the eligible people would actually join the new chapter? Be specific about your methods of gathering data.
-
A force of 1000 N is applied at point D on a machine shown in Figure Q3. Determine: (a) The reaction force at A (b) The force acting on hydraulic cylinder CE (c) The force acting on hydraulic...
-
Two trains, one traveling at 64.00 km/h and the other at 128.00 km/h, are headed toward one another along a straight, level track. When they are 739 m apart, each engine other's train and applies the...
-
At the beginning of the month, you owned $5,000 of General Dynamics, $4,000 of Starbucks, and $7,000 of Nike. The monthly returns for General Dynamics, Starbucks, and Nike were 7.50 percent, -1.66...
-
This assessment covers the following LOs. LO1: Adapt information systems to strategically achieve organisational goals and be able to design, develop and manage IT systems implementation to achieve...
-
Conch Republic Electronics is a midsized electronics manufacturer located in Key West, Florida. The company president is Shelly Couts, who inherited the company. The company originally repaired...
-
Use the following data to answer the next two (2) questions: Product 1 Product 2 Product 3 Direct Material Cost $25,000 $30,000 $35,000 Direct Labor Cost $30,000 $40,000 $50,000 Direct Labor Hours...
-
How might the structure of DDT be modified to make it again effective against resistant insects?
-
Explain which product is formed when each of these alkyl chlorides reacts with sodium ethoxide in ethanol.
-
Explain which of these alkyl chlorides reacts faster with sodium ethoxide in ethanol.
-
41 Consider the following two mutually exclusive projects: Year Cash Flow (X) Cash Flow (Y) -$ -$ 0 20,500 20,500 1 8,975 10,350 2 9,350 7,925 3 8,925 8,825 Calculate the IRR for each project. (Do...
-
Walt Disney expects to receive a Mex$ 1 6 million theatrical fee from Mexico in 9 0 days. The current spot rate is $ 0 . 1 3 2 1 / Mex$ , and the 9 0 - day forward rate is $ 0 . 1 2 4 2 / Mex$ . ( A...
-
You are given the following information for Wine and Cork Enterprises ( WCE ) : rRF = 4 % ; rM = 8 % ; RPM = 4 % , and beta = 1 . 2 What is WCE's required rate of return? Do not round intermediate...

Study smarter with the SolutionInn App


