Identify the different kinds of carbonyl functional groups in the following molecules: (a) COH O || LOCCH3
Question:
Identify the different kinds of carbonyl functional groups in the following molecules: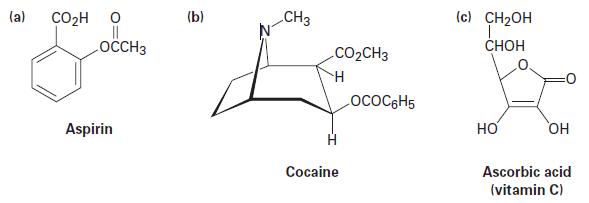
Transcribed Image Text:
(a) CO₂H O || LOCCH3 Aspirin (b) CH3 CO₂CH3 H Cocaine LOCOC6H5 (c) CH₂OH T CHOH НО OH Ascorbic acid (vitamin C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
In this problem we have to identify the type of carbonyl functional groups Our molecules are If our ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Many compounds contain more than one functional group. The structure of prostaglandin E1, a hormone that regulates the relaxation of smooth muscles, contains two different kinds of carbonyl groups....
-
Many compounds contain more than one functional group. The structure of prostaglandin E1, a hormone that regulates the relaxation of smooth muscles, contains two different kinds of carbonyl groups....
-
Identify the kinds of carbonyl groups in the following molecules (red=O, blue=N): (a) (b)
-
The boundedness theorem shows how the bottom row of a synthetic division is used to place upper and lower bounds on possible real zeros of a polynomial function. Let P(x) define a polynomial function...
-
Assume that Congress announces its intention to limit the prices and profits of pharmaceutical companies as part of an effort to control health care costs. What effect would you expect this...
-
(a) If each vertical gray line in Figure \(32.4 b\) represents \(0.020 \mathrm{~s}\), what is the frequency of the oscillation? (b) What is the average potential difference across the capacitor in...
-
The balance sheet of Hubble Ltd as at 31 May 2010 is shown below. Hubble Ltd agreed to purchase at this date the freehold premises, plant and machinery and stock _ of A Bubble at agreed valuations of...
-
The Davidson Corporation's balance sheet and income statement are provided here. a. Construct the statement of stockholders' equity for December 31, 2008.b. How much money has been reinvested in the...
-
H. 5 8 67% PART 2 Calculate the amount that Jaxson Pty Ltd could potentially distribute to its shareholders in a new distribution by the end of the current income year without incurring Franking...
-
What product would you expect from nucleophilic addition of methanol, CH 3 OH, to benzaldehyde under acidic conditions?
-
Compounds called cyanohydrins result from the nucleophilic addition of HCN to an aldehyde or ketone. Draw and the carbonyl compound that the following cyanohydrin was prepared from (red=O, blue=N):
-
Go to irs.gov and find the most recent IRS Publication 1542, Per Diem Rates. What is the maximum per diem rate for lodging and meals and incidental expenses (M&IE) for each of the following cities?...
-
Consider two firms, Zoe Corporation and Marley Company. Both corporations will either make $20,000 or lose $5,000 every year with equal probability. The firms' profits are perfectly negatively...
-
What is an IPO over-allotment (Greenshoe) option? Describe the over-allotment option included in Prada's IPO?
-
Question 2: Your pension fund has a sub-portfolio of bonds. The duration of this bond portfolio is 8 years. The current market value of the bond portfolio is $1,000,000. Calculate the price change...
-
A 286-room upscale hotel supplies each guestroom with a 1 oz. bar of facial soap, packed 288 per case, and a 2 oz. bar of bath soap, packed 144 per case. The ending inventory for the month of...
-
A small clinic in a small town treats only 10 patients. It expects operating cost per patient will be $1,800, or total $18,000. It also desires an operating margin of $3600. If the clinic treats one...
-
What is value-chain analysis? Explain the difference between primary and secondary activities.
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
In the following reaction, determine whether the alkyne has been oxidized, reduced, or neither. Using the answer from the previous problem, try to determine the answer without calculating oxidation...
-
While 13 C is the main contributor to the (M+1) + peak, there are many other elements that can also contribute to the (M+1) + peak. For example, there are two naturally occurring isotopes of...
-
Below are mass spectra for four different compounds. Identify whether each of these compounds contains a bromine atom, a chlorine atom, or neither. a. b. c. d. 100- 80- 60- 60- 40 20- 0- 60 70 10 20...
-
The flexible budget variance for fixed overhead is known as the A.) Static budget variance. B.) Fixed overhead efficiency variance. C.) The fixed overhead volume variance. D.) The fixed overhead...
-
Examine the balance sheet for MNO Ltd. as of December 31, 2023, to assess its financial performance and efficiency. MNO Ltd. Balance Sheet As of December 31, 2023 Assets Current Assets Cash $30,000...
-
P dx x tan 5x 2x tan 5x+5x2 sec 5x tan 5x O 2x tan 5x+5x2 sec 5x 10x sec 5x O 2x tan 5x + x2 sec 5x O 2x tan 5x + 5x2 sec 5x tan 5x

Study smarter with the SolutionInn App


