Name the following alkyl halides: (a) H3C Br Br | | || CH3CHCHCHCHCHCH3 CH3 (d) CHBr CH3CHCHCHCHCH3
Question:
Name the following alkyl halides:
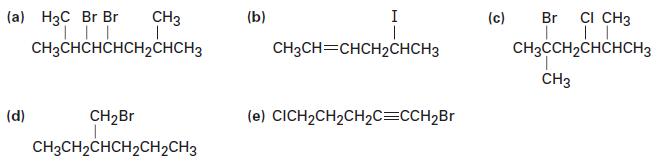
Transcribed Image Text:
(a) H3C Br Br | | || CH3CHCHCHCH₂CHCH3 CH3 (d) CH₂Br CH3CH₂CHCH₂CH₂CH3 (b) I CH3CH=CHCH2CHCH3 (e) CICH₂CH₂CH₂C=CCH₂Br (c) Br CI CH3 | || CH3CCH₂CHCHCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a 1bromo22dimethylpropane b 2bromo3methyl pentane c 3bromo1chlorobutane d 2bromo...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give a JUPAC name for each of the following alkyl halides (yellow green =Cl): (b) (a)
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
Tell which of the following alkyl halides can give only one alkene, and which can give a mixture of alkenes, in the E2 reaction. (a) (b) CH,CH,CHCH,Br CH3
-
Discuss why the length of an OH bond obtained from X-ray diffraction experiments averages 85 pm whereas that obtained in neutron diffraction experiments averages 96 pm. Would you expect to see...
-
Describe how a manager who derives satisfaction from both income and shirking allocates a 10- hour day between these activities when paid an annual, fixed salary of $ 100,000. When this same manager...
-
Seacrest Manufacturing produces ceramic teapots. Seacrest allocates overhead based on the number of direct labor hours. The company is looking into using a standard costing system and has developed...
-
Adaris Group, headquartered in the UK, owns a 70 percent interest in Adaris Malaysia. The acquisition cost was 200 million. At the date of acquisition, 1 January 2012, Adaris Malaysia's book value...
-
Premiums Presented below are three independent situations. 1. Mar-quart Stamp Company records stamp service revenue and provides for the cost of redemptions in the year stamps are sold to licensees....
-
Connor Ward wishes to have 1,100,000 in a retirement fund 25years from now. He can create the retirement fund by making asingle lump sum deposit today 0 answers
-
Draw structures corresponding to the following names: (a) 2-Chloro-3,3-dimethyl hexane (b) 3,3-Dichloro-2-methyl hexane (c) 3-Bromo-3-ethyl pentane (d) 2-Bromo-5-chloro-3-methylhexane
-
Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? (a) Br CH3 | | CH3CH2CHCHCH3 (b) CH3 CI CH3 | | CH3CHCH2-C-CHCH3 CH3...
-
How will you prepare the workforce to deal with the conversion to the lean shop?
-
Your investment advisor promises "invest $10,000 with me today and I double your money in four years." What is the implied interest rate (APR, annually compounded) in her promise?
-
The images above show agricultural activities that occur in the area surrounding a central city in the Middle East. Use your knowledge of agricultural land-use patterns to predict the location of...
-
(1 point) Consider a forward contract on a commodity with a current price of $2800 and delivery time in 4 months. Assume that the risk-free rate of interest is 7% compounded monthly and there is no...
-
Nano Limited manufactures two products. The production of Product 1 is entirely carried out in the Green department. The entire production of Product 2 takes place in the Yellow department. Nano...
-
Course: Strategic pricing decision Good evening, please your support with this question according to the data of the case: Case: McDONALD'S: THE HAMBURGER PRICE WARS...
-
What is an embargo? Explain why it is seldom used today.
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
Identify the stronger base: a) NaOH vs. H 2 O b) Sodium ethoxide vs. ethanol c) Ammonia vs. trimethylamine
-
(2S, 3S)-2-Bromo-3-phenylbutane undergoes an E2 reaction when treated with a strong base to produce (E)-2- phenyl-2-butene. Use Newman projections to explain the stereo-chemical outcome of this...
-
Department F had 4,000 units in work in process that were 40% completed at the beginning of the period at a cost of $14,100. During the period, direct materials were added at a cost of $26,500, and...
-
During an IT audit, the auditor finds that loan customers are unable to log in to their online profiles to correct information in an application. Which privacy principle is most affected? Disclosure...
-
Select all that apply On 2/1, Sophie Company sold merchandise on account to Lexie Company in the amount of $1,500. Lexie Company paid the amount due on 2/28. The journal entry that Sophie Company...

Study smarter with the SolutionInn App


