One enantiomer of lactic acid is shown below. Is it R or S? Draw its mirror image
Question:
One enantiomer of lactic acid is shown below. Is it R or S? Draw its mirror image in the standard tetrahedral representation.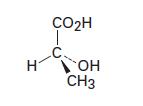
Transcribed Image Text:
CO2H С. Н он CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The lowest priority substituent H is not on a hatched wedge so make an int...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the tetrahedral representation of the S enantiomer of the amino acid cysteine, HSCH2CH(NH2)CO2H.
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Determine whether each function is one-to-one. If it is, find the inverse. f(x) = 4x - 1
-
You are the manager of a firm that produces products X and Y at zero cost. You know that different types of consumers value your two products differently, but you are unable to identify these...
-
Some have argued that social entrepreneurship is another form of commercial entrepreneurship with positive social or environmental change as its product. Do you agree with the accuracy of this...
-
Assume that Fleetwood Inc. purchased 40\% of the voting stock of Mac Corporation on January 1, 2020, for \(\$ 100,000\), an amount equal to \(40 \%\) of Mac's book value. Assume that the fair value...
-
(Prepared from a situation suggested by Professor John W. Hardy.) Lone Star Meat Packers is a major processor of beef and other meat products. The company has a large amount of T-bone steak on hand,...
-
Renfro Rentals has issued bonds that have a 9% coupon rate,payable semiannually. The bonds mature in 8 years, have a facevalue of $1,000, and a yield to maturity of 9%. What is the priceof the bond 2...
-
Tell whether the following Newman projection of 2-chlorobutane is R or S. H. H3C CI H CH3 H
-
Assign R or S configuration to the chirality centers in the following molecules: (a) HOH (b) CI H (c) H OCH3 HOCH2 CO2H
-
Determine whether the doubly infinite improper integral converges and, if so, evaluate it. Use definition (2). 00 J xdx 1 + x -00
-
1(x,0)=0 2x x! Given the Poisson distribution Confidence limits for the parameter 1. -,x=0,1,2... , obtain 100 (1-a )% "
-
Find the absolute maximum and absolute minimum of the function up above a) Find the corner points of the region. b) A critical point(s) that lie within the region. c) All the boundary points when...
-
Assume the CAPM is true. The risk - free rate is 5 % and the market risk premium is 7 % . What is the alpha of the market?
-
A company has an average inventory level of $ 2 , 8 6 4 , 0 0 0 , an average trade receivables level of $ 1 , 5 3 1 , 0 0 0 , and an average trade payables level of $ 7 2 6 , 0 0 0 . If the annual...
-
If 1 million people take a standardized test and Sarah scores two standard deviations above the mean, what percentile is she? 50% 68% 0 74% 85% O 97% Save Answer
-
Some people believe the rise of regional trading blocs threatens free trade progress made by the World Trade Organization (WTO). Do you agree? Why or why not?
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
Consider the following two isomers of 2,4-hexadiene. One isomer reacts rapidly as a diene in a Diels-Alder reaction, and the other does not. Identify which isomer is more reactive, and explain your...
-
Rank the following dienes in terms of reactivity in Diels- Alder reactions (from least reactive to most reactive):
-
Predict the products for each of the following reactions: (a) (b) (c) (d) (e) (f) NC CN CN NC
-
Under the partial equity method, a parent company Multiple select question. adjusts its share of the subsidiary reported income for intra-entity gross profits remaining in ending inventory. adjusts...
-
Current Attempt in Progress Ivanhoe Corporation reported the following information for 2021: Sales revenue $1013000 Cost of goods sold 709100 Operating expenses 111430 Unrealized holding gain on...
-
If total Debits in the account of cash equal $76,000 and the total credits equal $13,000, then the balance of cash is: O a. Debit $89,000 O b. Debit $63,000 c. Credit $63,000 O d. Credit $89,000

Study smarter with the SolutionInn App


