Propose structures for molecules that meet the following descriptions: (a) A ketone, C 5 H 10O (b)
Question:
Propose structures for molecules that meet the following descriptions: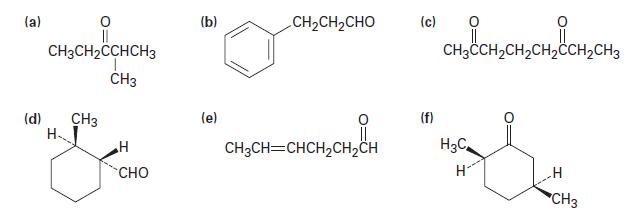 (a) A ketone, C5H10O
(a) A ketone, C5H10O
(b) An aldehyde, C6H10O
(c) A keto aldehyde, C6H10O2
(d) A cyclic ketone, C5H8O
Transcribed Image Text:
(a) (d) CH3CH₂CCHCH3 CH3 H CH3 H CHO (b) (e) -CH2CH,CHO CH3CH=CHCH₂CH₂CH (c) (f) CH3CCH₂CH₂CH₂CCH₂CH3 H3C H H CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Lets propose structures for molecules that meet the given descriptions a A ketone C5H10O A ketone ha...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose structures for molecules that meet the following descriptions. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 6 H 12 O; IR: 1715 cm 1 ; 13 C NMR: 8.0 ...
-
Propose structures that meet the following descriptions: (a) A ketone with five carbons (b) A four-carbon amide (c) A five-carbon ester (d) An aromatic aldehydes (e) A keto ester (f) An amino alcohol
-
Propose structures for molecules that meet the following descriptions: (a) Contains two sp2-hybridized carbons and two sp3-hybrized carbons (b) Contains only four carbons, all of which are...
-
Find each product. (8 m)( + m)
-
In this chapters Headline, we learned that Tom Jackson benefits by announcing well in advance his new marketing plan to target businesses. Suppose you are an executive at Mortons, and by lucky...
-
What are some initial frst steps that social entrepreneurs should be aware of as they embark on writing their social venture plan?
-
Explain why the amount of cash salaries paid to employees does not equal salaries expense for the employer.
-
The Pitts Barbecue Company makes three kinds of barbecue sauce: Extra Hot, Hot, and Mild. Pitts vice president of marketing estimates that the company can sell 8,000 cases of its Extra Hot sauce plus...
-
Question 6 John Adams plans to retire at the age of 62. He wantsan annual income of $60,000 per year. He believes that he will liveto be 87. He will draw the money at the beginning of each year. How...
-
Predict the products of the reaction of the following substances with CrO 3 in aqueous acid: (a) CH3CHCHCHCH (b) CH3 CH3CH,CH,CH, CHO CH3 (c)
-
Draw the product(s) obtained by conjugate addition of the following reagents to cyclohex-2-enone: (a) H 2 O (b) NH 3 (c) CH 3 OH (d) CH 3 CH 2 SH How can you explain the observation that the S N 2...
-
A box-shaped metal can has dimensions 8 in. by 4 in. by 10 in. high. All of the air inside the can is removed with a vacuum pump. Assuming normal atmospheric pressure outside the can, find the total...
-
G & are shown below. G = 30, & N = 19. Let R = G-. Determine the components of each vector as well as the magnitude & direction of R. For hardcopies of this assignment, you can draw R in the blank...
-
1)Why Shareholder equity is consider the liability of the company? 2)Explain the difference between Income Statement and Cash Flow Statement? 3)Why Company do not like to issue Preferred Stock? and...
-
Your investment portfolio has two funds in it. Fund A provides 13% return a year with risk of 19%. Fund B provides 8% a year with risk of 11%. The two funds has a correlation coefficient of 0.4. If...
-
AMAT currently pays $1.8 in dividend. The company can increase dividend at 10% a year. If the required return is 12.5%, what should be the stock price for AMAT in 10 years?
-
Evaluate the limit Answer: L = 5 5n 2 2 2 /25 - (- 5 + 57) . N L = lim N0 # Hint: Here each term being added depends on the number of terms involved. Therefore this problem cannot be addressed by our...
-
What is the international equity market? Identify the factors responsible for its expansion.
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
For each of the following compounds, compare the two indicated protons and determine whether they are enantiotopic, homotopic, or diastereotopic: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m)...
-
Draw the expected 1 H NMR spectrum of the following compound: CI
-
Consider the following compound: (a) How many signals do you expect in the 1 H NMR spectrum of this compound? (b) Rank the protons in terms of increasing chemical shift. (c) How many signals do you...
-
Please answer with reason for all why the option is correct and why the other options are incorrect,, Please help with this Accounting type Question: The current ratio is ratio between: A . Current...
-
m(x) = csc(3x) - 1 is increasing on (, ) ...()u(0)(.) U... ...U (3,0) U(0, 1) U ( 3 O...U()()()u... U ... () (0) (,)..... U 3 none
-
The general environment analysis considers broad societal dimensions influencing the industry of firms within it. The general environment areas include? A. Political, Economic, Social, Technical,...

Study smarter with the SolutionInn App


