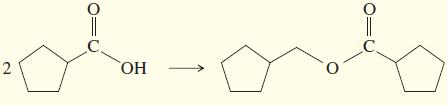
Show how you would accomplish the following multistep syntheses. You may use any additional reagents and solvents
Question:
Show how you would accomplish the following multistep syntheses. You may use any additional reagents and solvents you need.
a. PhCH2CH2OH → PhCH2CH2COOH
b.
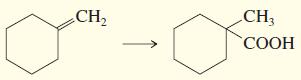
c.
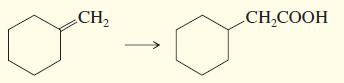
d.
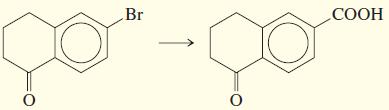
e.
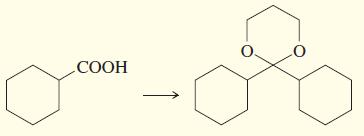
f.
Transcribed Image Text:
CH3 CH2 СООН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b c d e f Ph...View the full answer

Answered By

AKASH PANJA
I'm currently in 4th semester of Msc organic chemistry. I'm doing MSc from Central University Of Haryana, India. I've done two research Projects from Centre Of Biomedical Research (CBMR), Lucknow, India in organocatalysis and metal Catalyzed Asymmetric synthesis.
I'm a former subject expert in chegg. I can solve any questions of chemistry with a very detailed explanations in a cheap rate compare to others.
https://www.linkedin.com/in/akash-panja-516247196
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you would accomplish the following synthetic conversions. You may use any additional reagents and solvents you need. (a) (b) (c) (d) Ph-CHO OH PhCHOPhCHC CCH2CH3
-
Show how you would accomplish the following multistep conversions. You may use any additional reagents you need. a. b. c. d. dimethyl adipate and allyl bromide
-
Show how you would accomplish the following syntheses. You may use whatever additional reagents you need. (a) (b) (c) (d) (e) (f) CHO CH,OH CHO CHO CH,Br Ph of CH, CH cth CH CH CHC-Ph 0 BrCH,CH,CCH...
-
Use any method to determine if the series converges or diverges. Give reasons for your answer. n10 10" n=1
-
Find [Cu2+] in a solution saturated with Cu4(OH)6(SO4) if [OH-1] is fixed at 1.0 10-6M. Note that Cu4(OH)6(SO4) gives 1 mo of SO42- for 4 mol of Cu2+. Ksp = 2.3 10-69
-
Explain the basic principles of double-entry bookkeeping.
-
A UK company hedges its U.S. dollar purchase of equipment from a U.S. supplier using a forward contract exchanging pounds for U.S. dollars at a fixed rate. The forward qualifies as a hedge of a...
-
The information listed below refers to the employees of Lemonica Company for the year ended December 31, 2016. The wages are separated into the quarters in which they were paid to the individual...
-
MC Qu. 63 The times interest earned... H newconnectmheducationcom Exam 3B (40) G Help Save 5 Exit Submit 6 MC On. 63 The times Interest earned... The times interest earned ratio reflects: Multiple...
-
A cast iron, T-type fitting is being produced for the oil drilling industry, using an air-set or no-bake sand for both the mold and the core. A silica sand has been used in combination with a...
-
When pure (S)-lactic acid is esterified by racemic butan-2-ol, the product is 2-butyl lactate, with the following structure: (a) Draw three-dimensional structures of the two stereoisomers formed,...
-
The following NMR spectra correspond to compounds of formulas (A) C 9 H 10 O 2 , (B) C 4 H 6 O 2 , and (C) respectively. Propose structures, and show how they are consistent with the observed...
-
Heat in the amount of 100 kJ is transferred directly from a hot reservoir at 1200 K to a cold reservoir at 600 K. Calculate the entropy change of the two reservoirs and determine if the increase of...
-
List and explain the three theories for why the SRAS curve is upwards sloping.
-
Explain why the LRAS curve is vertical.
-
How might a government use taxation as a tool in expansionary fiscal policy?
-
Explain why, in the short run, there might be a trade-off between inflation and unemployment.
-
Why do flexible labour markets improve the efficiency and capacity of the economy?
-
A retailer's store is destroyed by a tornado, but is insured for its replacement cost. Consequently, the retailer has a $40,000 gain after receiving the insurance proceeds. The store is not replaced...
-
Write a paper detailing a geographic information system (GIS) of your own design that would utilize data in an original manner.
-
Propose a curved-arrow mechanism for the following reaction. Explain why the equilibrium lies to the right. Ph Ph CH toluenesulfonic acidCH
-
Why is trityl chloride much more reactive than the other alkyl halides in Table 17.2? TABLE 17 2 comparison of S,1 Solvolysis Rates of Benzylic and Nonbenzylic Alkyl Halides 25C R-CIHo l + H2O -OH +...
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CC14 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
Read the scenario located in "Problems - Series A," section 8-19A of Ch. 8, "Performance Evaluation," of Fundamentals of Managerial Accounting Concepts . This scenario puts you in charge of pr...
-
F of 2 Book Hint Required information Use the following information for the Exercises 3-7 below. (Algo) [The following information applies to the questions displayed below] Laker Company reported the...
-
Anita Strods opened a business called Strods Engineering and recorded the following transactions in its first month of operations. June 1 Anita Strods, the owner, invested $156,000 cash, office...

Study smarter with the SolutionInn App


