Tetracaine, a substance used as a spinal anesthetic, can be prepared from benzene by the following route.
Question:
Tetracaine, a substance used as a spinal anesthetic, can be prepared from benzene by the following route. Show how you could accomplish each of the transformations (a) through (d).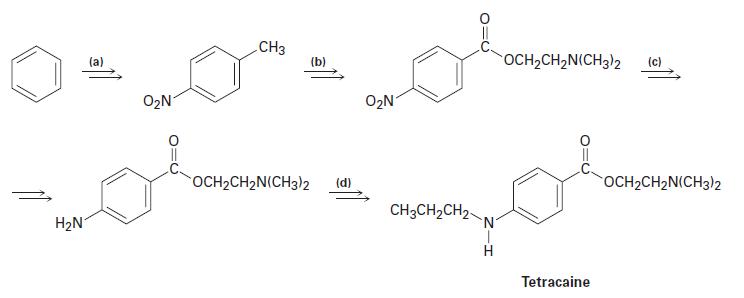
Transcribed Image Text:
H₂N (a) O₂N CH3 (b) OCH₂CH₂N(CH3)2 (d) O₂N CH3CH₂CH₂- N OCH₂CH₂N(CH3)2 Tetracaine OCH₂CH2N(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
nhn nhiu nhiu nhiu c nhiu chu nhiu ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you could prepare each of the following amines from benzaldehyde by reductive amination: (a) Benzylamine (c) N, N-Dimethylbenzylamine (b) Dibenzylamine (d) N-Benzylpiperidine
-
Show how you could prepare each of the following compounds from cyclopentanone, D2O, and any necessary organic or inorganic reagents. H OH
-
Show how you could prepare each of the following compounds. Use the starting material indicated along with ethyl acetoacetate or diethyl malonate and any necessary inorganic reagents. Assume also...
-
In Exercises 1126, determine whether each equation defines y as a function of x. 4x = y 2
-
Jay Maxey retired a few years ago at age 48, courtesy of the numerous stock options he had been granted while president of e-shops.com, an Internet start-up company. He soon moved to Montana to...
-
At the instant shown in Figure 32. 40 , the potential difference across the capacitor is half its maximum value and the charge on the plates is increasing. Draw the direction of the current and...
-
\(\frac{3}{8} \times\left(\frac{13}{12}-\frac{35}{36} ight)\) Perform the indicated operations. Reduce to lowest terms.
-
Stainless Refrigerator Company purchases ice makers and installs them in its products. The ice makers cost $138 per case, and each case contains 12 ice makers. The supplier recently gave advance...
-
Q3. (22 marks) Adam Hamshere, the ?nance director of Washington Oil Investments Ltd (WOIL), is unsure whether he should consolidate some of the investments that the company owns. He has asked your...
-
How might you use a reductive amination to synthesize ephedrine, an amino alcohol that is widely used for the treatment of bronchial asthma? CHCHNHCH3 CH3 Ephedrine
-
The amino acid proline is biosynthesized from glutamate semialdehyde by the following transformation, where NADH is the biological reducing agent nicotinamide adenine dinucleotide. What is the likely...
-
Describe the four phases that go into developing a business model.
-
QUESTION 1 71/76 Sam Hill has just won the state lottery, paying $250,000 a year for the next 30 years. Sam will receive his first payment today. If the interest rate is 8%, what is the present value...
-
A. Look at the capital structures of eBay and Google. Can you identify any reasons for the difference? Can you think of any comments or recommendations? Does any of the capital structure theories fit...
-
Quetzaltenango Candle Inc. budgeted production of 740,000 candles for March. Wax is required to produce a candle. Assume 11 ounces of wax is required for each candle. The estimated March 1 wax...
-
An apartment building owner has a mortgage balance of $2,465,000. He also owes $22,575 in back-owed property taxes (plus penalties of 10%) and a foreclosure trustee's sale has been set - so he is...
-
Number of items Unit price (5 $5.00, 7 $8.00, 8 $6.00, 12 $3.00) Purchases of an inventory item during last month are summarized in the brackets. What was the weighted average price per item?
-
You are interested in home building trends in the United States as your company, Whirlpool, is a major supplier of kitchen appliances. Go to www.nahb.com and describe what types of information at...
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Identify the reagents necessary to accomplish each of the following transformations. If you are having trouble, the reagents for these transformations appear on page 482, but you should first try to...
-
Identify the reagents you would use to accomplish each of the following transformations: a. b.
-
Identify the reagents you would use to accomplish each of the following transformations: a. b.
-
A tax on sellers will shift the Question 5 0 options: Demand curve to the right. Supply curve to the right. Demand curve to the left. Supply curve to the left. Equilibrium quantity to the right.
-
P 1 was $ 2 0 , Q 1 was 1 0 . The seller raised price to P 2 = $ 2 5 and Q 2 became 9 . Demand in this case is Question 3 2 options: Elastic Inelastic Perfectly elastic Perfectly inelastic Unit...
-
RiverRocks, Inc., is considering a project with the following projected free cash flows: Year Cash Flow (in millions) 0 1 2 3 4 - $49.6 $9.6 $19.4 $19.2 $15.6 The firm believes that, given the risk...

Study smarter with the SolutionInn App


