Which of the following reactions will occur? (For the pK a values necessary to do this problem,
Question:
Which of the following reactions will occur? (For the pKa values necessary to do this problem, see Appendix II.)
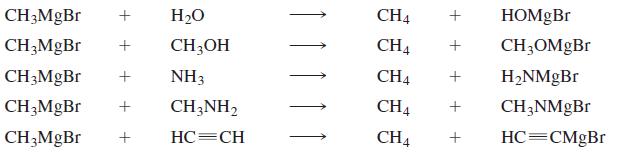
Transcribed Image Text:
H20 CH4 HOMGB. CH;OH CH4 + CH3OMGBR CH3MGB NH3 CH4 + H2NMgBr CH3MGB CH3NH2 CH4 CH;NMGB CH;MgBr + HC=CH CH4 HC=CMgBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Grignard reagent has very high propensity to capture the ...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions will go faster if the concentration of the nucleophile is increased? a. b. c. H. Br H OCH Br Br SCH3 Br OCCH3 + Br
-
Which of the following reactions is more likely to occur?Explain. eat eat 2.
-
Which of the following reactions occurs most rapidly? Why? a. b. c. Br - H20 C(CH3)3 C(CH3)a (CH)a C(CH3)3 Br - H20 CH33 C(CH3)3
-
Suppose that a telephone switchboard of some company on the average handles 300 calls per hour, and that the board can make at most 10 connections per minute. Using the Poisson distribution, estimate...
-
Consider a planar waveguide used for attenuated total reflection measurement of a film coated on one surface of the waveguide. For a given angle of incidence, the sensitivity of attenuated total...
-
The 2014 financial. statements of LVMH Moet Hennessey-Louis Vuitton S.A. are presented in Appendix C of this book. LVMH is a Paris-based holding company and one of the worlds largest and best-known...
-
A lessor, Ace Corp. enters into an equipment lease with a lessee, Spades Inc. The terms of the lease require annual lease payments of \(\$ 48,000\) over a 10 -year period, with the first payment due...
-
Turner Excavation maintains a checking account and has decided to open a petty cash fund. The following petty cash fund transactions occurred during July: July 2 Established a petty cash fund by...
-
How much are you willing to pay for one share of Newman Inc. stock if the company just paid a $1.75 annual dividend, the dividends increase by 4 percent annually, and you require a 9.6 percent rate...
-
You are appraising a single-family residence located in the Huntington neighborhood at 4632 NW 56th Drive. The property is being acquired by a mortgage applicant and you have been asked to appraise...
-
Show, using any necessary reagents, how the following compounds could be prepared with ethylene oxide as one of the reactants: a. CH 3 CH 2 CH 2 CH 2 OH b. CH 3 CH 2 CH 2 CH 2 D c. CH 3 CH 2 CH 2 CH...
-
Explain why tertiary alkyl halides cannot be used in coupling reactions with Gilman reagents.
-
Why is maintaining the entanglement of pairs A and B and A and X the crucial ingredient of teleportation?
-
Use the Psychsocial Systems Model as this will be the model you will use to analyze this case. follow the guideline as listed : 1. Introduction 2. Leadership Assessment 3. The Contextual Factors in...
-
Solve the following differential equation: 1. y'" 2y" + y' =2-24e* - 40e5*, y(0) = 0.5, y'(0) = 2.5, y"(0) = -4.5
-
By using this article www.ncbi.nlm.nih.gov/pmc/articles/PMC5032978/ you have to attend the following question's answer and also must include the citation. Identify the specific the type of data that...
-
Compensation data for numerous jobs are more available due to the existence of various websites. The website of Salary.com provides pay data for numerous jobs in different companies in different...
-
Imagine that you are the supervisor of the transcription group of the health information services department at Community Hospital. Your crew consists of several transcriptionists who handle, in...
-
What is a production cost report, and how is it used by management?
-
Suppose that A is an m n matrix with linearly independent columns and the linear system LS(A, b) is consistent. Show that this system has a unique solution.
-
When compound A is irradiated with ultraviolet light for 115 hours in pentane, an isomeric compound B is obtained that decolorizes bromine in CH2C12 and reacts with ozone to give, after the usual...
-
Heptafulvalene undergoes a thermal reaction with tetracyanoethylene (TCNE) to give the adduct shown in Fig. P27.36. What is the stereochemistry of this adduct? Explain. NC CN NC CN heptafulvalene NC...
-
Explain why the following equilibrium lies far to the right. CH2 CH toluene (B)
-
1. Provide an explanation on Audit of Performance Information in the Public Sector.(Advanced Audit and Assurance) 2. What areSocial and Environmental Issues.(Advanced Audit and Assurance)
-
9 On January 1, 2024, Riverbed Company acquired all the assets and assumed all the liabilities of Oriole Company and merged Oriole into Riverbed. In exchange for the net assets of Oriole, Riverbed...
-
Part I. Recording the transactions. Exercise 1. Blue Co engaged in the following transactions during July 2022: Required: Based on the information below, journalize the transactions. (36 Points) July...

Study smarter with the SolutionInn App


