Arrange the following compounds according to increasing rate of elimination with NaOEt in EtOH. What is the
Question:
Arrange the following compounds according to increasing rate of elimination with NaOEt in EtOH. What is the product in each case?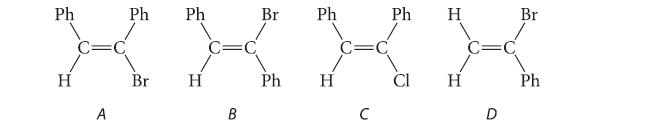
Transcribed Image Text:
Ph H C=C A Ph Br Ph H C=C B Br Ph Ph H C=C C Ph Cl H H =C D Br Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Assume that E2 reactions of vinylic halides follow the same trends as E2 reactions of alkyl h...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following compounds in order of increasing boiling point. Explain your answer in terms of the intermolecular forces in each compound. (a) (b) (c) (d)
-
Arrange the following compounds in order of increasing reactivity toward HNO3 in H2SO4. (The references to equations will assist you with nomenclature.) (a) Chlorobenzene, benzene, nitrobenzene (b)...
-
Arrange the following compounds in order of increasing boiling point. (b) (a) (d) (c)
-
Determine whether the following code fragment takes linear time, quadratic time, or cubic time (as a function of \(n\) ). for (int i = 0; i < n; i++) for (int j = 0; j < n; j++) j) C[i][j] 1.0; if (i...
-
The proper mean lifetime of pions is 2.6 10-8 s. If a beam of pions has a speed of 0.85c, (a) What would their mean lifetime be as measured in the laboratory? (b) How far would they travel, on...
-
A solid round bar of aluminum having diameter d (see figure) is compressed by an axial force P = 60 kN. The bar has pinned supports and is made of alloy 6061-T6. (a) If the diameter d = 30 mm, what...
-
If you were stranded on a deserted island with only one criterion for measuring value creation, which would you want to use? Why?
-
Do you believe Michael Woodford did the right thing by blowing the whistle on accounting irregularities? Were there other options open to him? Once he was fired, could he have made a whistleblower's...
-
y = 2 tan () Drag either of the movable red points to set the asymptotes for one period of the given function. Also place the blue point at the correct set of coordinates. You may click on a point to...
-
Within each set, rank the compounds in order of increasing rates of their S N 2 reactions. Explain your reasoning. (a) Benzyl bromide, (3bromopropyl) benzene, pbromotoluene (b) 1bromocyclohexene,...
-
Consider the following mechanism for Eq. 18.19. Identify the process associated with each step. Counting electrons at each stage may help you. Pd(PPH3)4 Ph3P Ph,P Pd(PPh 3)2 + Pd H Br Ph H PhP...
-
Quality Shoe Company is considering investing in one of two machines that attach heels to shoes. Machine A costs \(\$ 60,000\) and is expected to save the company \(\$ 18,000\) per year for six...
-
1 . Calculate the allocation rate. Allocate the operating costs using cost of goods sold as the allocation base and compute the operating income of each market segment. 2 . Use the activity - based...
-
What is the lower-of-cost-or-net-realizable-value (LCNRV) of the ending inventory? Inventory NRV Cost Categories Street bikes Data Data $11,000 $10,200 Racing bikes 8,000 8,500 Mountain bikes 14,000...
-
Blossom Corp. has collected the following data concerning its maintenance costs for the past six months: Units Produced Total Cost July 17,500 $31,232 August 32,000 48,800 September 36,700 55,700...
-
What methods can be used to engage various stakeholders in a business setting, and how can communication be tailored to meet the needs and expectations of different stakeholder groups without...
-
How do seasonal trends or holidays impact consumer behavior in our industry, and how should we adjust our marketing calendar accordingly? Explain in detail.
-
For each item on the following list, give a reasonable method of sterilization. You cannot use the same method more than three times for the whole list. The method must sterilize, not just disinfect;...
-
Describe the Operations (+,,*,/) that can cause negligible addition (NA), error magnification (EM), or subtractive cancellation (SC) in calculating ?((x^2)+1) - x . Give the range of where they might...
-
Determine whether the following objects are chiral or achiral. T 0/
-
If you had the two enantiomers of carvone in unmarked bottles, could you use just your nose and a polarimeter to determine? (a) Whether it is the (+) or (-) enantiomer that smells like spearmint? (b)...
-
When optically pure (R)-2-bromobutane is heated with water, butan-2-ol is the product. The reaction forms twice as much (S)-butan-2-ol as (R)-butan-2-ol. Calculate the e.e. and the specific rotation...
-
1. Which of the following is NOT authoritative guidance that applies AICPA members providing forensic accounting services? a. Generally accepted auditing standards b. AICPA Code of Professional...
-
Select all that apply Not recording an accrued expense will have the following effect on the financial statement: Multiple select question. expenses on the income statement will be overstated....
-
If Total Liabilities increase, which of the following MUST be true?Group of answer choicesAssets MUST increaseAssets minus owners equity Must increaseOwners equity MUST increaseOwners equity minus...

Study smarter with the SolutionInn App


