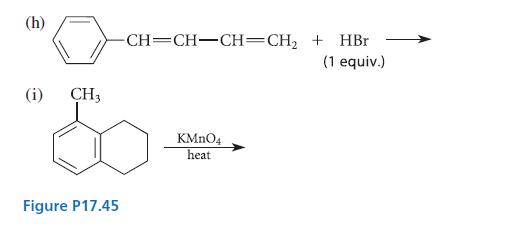
Complete the reactions given in Fig. P17.45 by proposing structures for the major organic products. (a) HC=CH-CH=CH-CHMgBr
Question:
Complete the reactions given in Fig. P17.45 by proposing structures for the major organic products.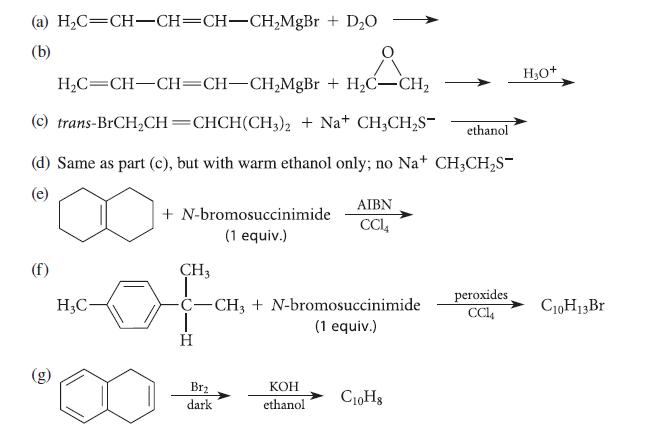
Transcribed Image Text:
(a) H₂C=CH-CH=CH-CH₂MgBr + D₂O (b) + H₂CH₂ H₂C=CH-CH=CH-CH₂MgBr + H₂C-CH₂ (c) trans-BrCH₂CH=CHCH(CH3)2 + Na+ CH3CH₂S- (d) Same as part (c), but with warm ethanol only; no Na+ CH3CH₂S- (e) (f) H₂C- + N-bromosuccinimide (1 equiv.) CH3 | C-CH3 + N-bromosuccinimide (1 equiv.) T H Brz dark AIBN CCL4 KOH ethanol ethanol C10H8 peroxides CCl4 H3O+ C10H13 Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
b Two allylic Grignard reagents are in equilibrium and each reacts with DO HCCHCHCHCHMgBr HCCHCHCHCH DO HCCHCHCHCHD MgBr DO HC CHCHCHCH D Note in this ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
Complete the reactions given in Fig. P19.45 by giving the principal organic product(s). Fig. P19.45 (a) (b) (c) p-toluenesulfonic acid (catalyst CH, t CH,OH - (solvent) ether Hio -caphenone, +...
-
Complete each of the reactions given in Fig. P20.49 by giving the principal organic product(s). Give the reasons for your answers. H,C KOH PhCH Cl acid HO,C CO,H+ethylene glycolheolym) CH + Hg(OAch...
-
The reconciling item in a bank reconciliation that will result in an adjusting entry by the depositor is: (a) outstanding checks. (b) deposit in transit. (c) a bank error. (d) bank service charges.
-
The radius of the orbit of a charged particle in a magnetic field is related to the momentum of the particle by p = BqR 39-41 This equation holds classically for p = mu and relativistically for p = m...
-
A tall standpipe with an open top (see figure) has diameter d = 2.2 m and wall thickness t = 20 mm. (a) What height h of water will produce a circumferential stress of 12 MPa in the wall of the...
-
The standard deviation of the earnings on Bouygues shares is 40%, while for Siemens it is only 28%. However, Bouygues has a of 1.13 and Siemens of 1.7. Explain how this is possible.
-
Bigelow Corporation has total assets of $850,000, sales of $1,350,000, and one class of common stock with 375 shareholders, and a class of preferred stock with 250 shareholders, both of which are...
-
You have conducted a risk analysis to protect a key company asset. You identify the following values: Asset value = 5000 Exposure factor = 50 Annualized rate of occurrence = .10 What is the...
-
Propose a curved-arrow mechanism for the reaction given in Fig. P17.48, and give at least two structural reasons why the equilibrium lies to the right. CH - CH=CH Figure P17.48 H CH3 resistanc H...
-
Account for each of the following facts with an explanation. (a) 1,3-Cyclopentadiene is a considerably stronger carbon acid than 1,4-pentadiene, even though the acidic hydrogens in both cases are...
-
In problem, x = 0 is a regular singular point of the given differential equation. Show that the indicial roots of the singularity differ by an integer. Use the method of Frobenius to obtain at least...
-
A capillary tube has a diameter of 1,65 mm and a height of 97.1 mm. What is the volume of the capillary tube in meters? (4 pts) 2. Convert 79 lbs to kg (2 pt) Convert 95.2 C to K. (2 pt) 3. a. A gas...
-
SCENARIO: Make a report on the role of the construction manager. Discuss the roles, responsibilities, management techniques, behaviour and ethics, health, and safety at the workplace. From the above...
-
b. C. Trace the output of the following code fragment: for (int i 5; i >= 0; i--) { } = int j = 0; while (j i) { } cout < (i + 2)) { cout
-
Palma acquired 6 % of the ordinary shares in Pan Ltd , a trading company, in June 2 0 1 2 for 6 0 , 0 0 0 . She sold her shares in March 2 0 2 3 for 5 1 0 , 0 0 0 . Palma has worked for Pan Ltd for...
-
Teal Mountain Corporation had sales of $3,000,000 for the year ended December 31, 2025. The unit selling price was $12. In making the sales, the company incurred the following costs and expenses....
-
From chapter 20, figure 20.11a. What biochemical characteristic does this figure illustrate? How could this characteristic be used to begin the identification of the isolated bacteria in this...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
When 2-methyl-2-propenal is affected with allylmagnesium chloride (H2C=CH-CH2-MgCl) in ether, then with dilute aqueous acid, a compound A is obtained, which, when heated strongly, yields an aldehyde...
-
Using phelol and any other reagents as starting materials, outline a synthesis of each of the following compounds. OCH CH,CH,COH
-
Each of the following reactions involves a sequence of twa pericyclic reactions. Identify the intermediate X or I involved in each reaction, and describe the pericyclic reactions involved. light
-
Mint, now part of Intuit, entered the market in 2005 with comprehensive online money management systems that by aggregating financial information from online bank and credit card accounts tell users...
-
Which benefit has federal regulations that directly tie the physician's benefit to the employees' benefit (if offered to physician must also be offered to employee)? a. Flexible Spending Account...
-
Which of the following is a prohibited act under Chapter 673? Question 20Select one: a. To use a suspended license b. To prepare a fraudulent return c. To use the license of another person d. All of...

Study smarter with the SolutionInn App


