Consider the detailed structure of the semiquinone? QH shown in Eq. 18.79a. (a) There are two possible
Question:
Consider the detailed structure of the semiquinone? QH shown in Eq. 18.79a.
(a) There are two possible structures for this semiquinone; draw them both.
(b) Show the resonance structures for either of the structures you gave in part (a).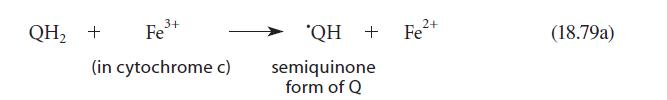
Transcribed Image Text:
QH₂ + 3+ Fe³+ (in cytochrome c) 'QH + Fe²+ semiquinone form of Q (18.79a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a CH3O CH30 0 CH3 H 10 or CH3O ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
There are two possible structures of XeF2Cl2, where Xe is the central atom. Draw them, and describe how measurements of dipole moments might be used to distinguish among them.
-
Consider the following reactions. For parts b-d, reference Exercise 58. a. When C5H12 is reacted with Cl2(g) in the presence of ultraviolet light, four different monochlorination products form. What...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Show that an emission tax and an absolute emission standard are equivalent instruments to regulate a polluting monopolist if and only if the standard is binding.
-
How fast must you be moving toward a red light ( = 650 nm) for it to appear green ( = 525 nm)?
-
A simple beam AB is subjected to a load in the form of a couple M0 acting at end B (see figure). Determine the angles of rotation θA and θB at the supports and the...
-
As a result of a change in the nature of its business, there is a relative rise in the proportion of fixed costs in a group As total costs. Will this affect the risk attached to its share price? If...
-
Shankar Company uses a perpetual system to record inventory transactions. The company purchases inventory on account on February 2, 2015, for $40,000, with terms 3/10, n/30. On February 10, the...
-
Transaction a. Balance Sheet Income Statement b. Statement of Cash Flows Total Assets Total Liabilities Total Net Equity Income 1. Owner invests $900 cash in business in exchange for stock +900 +900...
-
Draw the important resonance structures of the radicals formed when each of the following reacts with R?, a general free radical. (a) Vitamin E (b) BHT
-
(a) Using the fishhook notation, derive the important resonance structures of the vitamin C-derived radical in Eq. 18.81b. (b) In the laboratory, the radical derived from vitamin E can react with a...
-
In HUD/FHA settlement requirements, what rules apply to the handling of the down payment? To the handling of the closing costs?
-
Crosswinds Hospital plans to use activity-based costing to assign hospital indirect costs to the care of patients. The hospital has identified the following activities and activity rates for the...
-
Consider the following data for two products of Vigano Manufacturing. Activity Machine setup Parts handling Quality inspections Total budgeted overhead Unit Information Units produced Direct...
-
A simple, yet powerful model for spark ignition engines is the Otto cycle. In the ideal Otto Cycle, the spark, the fuel and the intake/exhaust stages are replaced with simpler stages. There are 4...
-
What key milestone is triggered when the project charter is signed?
-
A manufacturer uses normal costing and allocates manufacturing overhead costs to products on the basis of Machine hours in the Machining Department and on the basis of Direct labor hours in the...
-
a. What is the likelihood that plants have some sort of immune protection? b. Explain your reasoning.
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Draw the most stable conformation of: (a) ethylcyclohexane (b) 3-isopropyl-1,1-dimethylcyclohexane (c) cis-1-tert-butyl-4-isopropylcyclohexane
-
(a) Draw both chair conformations of cis-1,4-dimethylcyclohexane, and determine which conformer is more stable. (b) Repeat for the trans isomer. (c) Predict which isomer (cis or trans) is more stable.
-
Use your results from Problem 3-25 to complete the following table. Each entry shows the positions of two groups arranged as shown. For example, two groups that are trans on adjacent carbons...
-
YoY growth rate of 17 percent Plant currently operating at capacity Growth will require a new machine costing $3 million Assume additional $300,000 of annual depreciation for this new machine...
-
For this case, you will use selling price and cost data to determine if a special order from a customer should be accepted. Donut House wants to examine if it should accept a special order by a...
-
On 1 January 20.1 SA Traders purchased office furniture for R80 000. The financial year-end of SA Traders is 31 December.Calculate the accumulated depreciation for 20.3 by using the reducing-balance...

Study smarter with the SolutionInn App


