Nitration of phenyl acetate (compound A) results in para substitution of the nitro group. However, nitration of
Question:
Nitration of phenyl acetate (compound A) results in para substitution of the nitro group. However, nitration of dimethyl phenyl phosphate (compound B) results in meta substitution of the nitro group. Suggest a reason that the two compounds nitrate in different positions.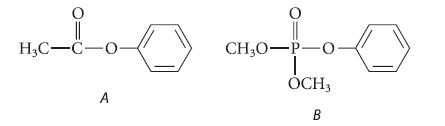
Transcribed Image Text:
H3C-C-O- A 0 CH3O-P-0 OCH3 B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
First draw the resonance structure of compound B that has an electronic octet around phosphorus This ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In an aqueous solution containing sodium bicarbonate, aniline reacts quickly with bromine to give 2, 4, 6-tribromoaniline. Nitration of aniline requires very strong conditions, however, and the...
-
Compounds A and B are carboxylic acids. Identify each one on the basis of its 1H NMR spectrum. (a) Compound A (C3H5ClO2) (Figure 19.10). (b) Compound B (C9H9NO4) has a nitro group attached to an...
-
Compounds A and B are carboxylic acids. Identify each one on the basis of its 1H NMR spectrum. (a) Compound A (C3H5ClO2) (Figure 19.10). (b) Compound B (C9H9NO4) has a nitro group attached to an...
-
Write a StockAccount client that builds an array of StockAccount objects, computes the total value of each account, and prints a report for the accounts with the largest and smallest values. Assume...
-
The combination of physical constants = e2k/hc, where k is the Coulomb constant, is known as the fine-structure constant. It appears in numerous relations in atomic physics. (a) Show that is...
-
The roof beams of a warehouse are supported by pipe columns (see figure on the next page) having outer diameter d2 = 100 mm and inner diameter d1 = 90 mm. The columns have length L = 4.0 m, modulus E...
-
Why do EVA adversaries describe it as a great marketing stunt?
-
Compute the amount that can be borrowed under each of the following circumstances: 1. A promise to repay $90,000 seven years from now at an interest rate of 6%. 2. An agreement made on February 1,...
-
Find the limit of the sequence: an = 2 2 1n2+8n+5 2n2+8n+6 n+8n+5 X syntax incomplete.
-
Suppose that the following curved-arrow notation and resonance structures for TPP ylid have been proposed in an attempt to demonstrate that the charges in the ylid can be neutralized by resonance....
-
According to the introduction to this chapter, trade models built exclusively on the idea of comparative advantage have a mixed record when it comes to predicting a countrys trade pattern. Using the...
-
Allow two-party competition in the elections over infinite time horizon, \(t=0,1, \ldots,+\infty\). Assume that there is an election every other period and two parties, \(i=L, R\) ("left" and...
-
The following is a record of Oriole Company's transactions for Boston Teapots for the month of May 2025. May 1 Balance 480 units @ $19.00 May 10 Sale 360 units @ $38.00 12 Purchase 720 units @ $25.00...
-
Assuming the user enters a and b as positive integers. The output of the program is a= int(input()) b= int(input()) q=a//b r=a%b if a ==(q*b+r): print("hi") else: print("bye")
-
Stanley-Morgan Industries adopted a defined benefit pension plan on April 12, 2024. The provisions of the plan were not made retroactive to prior years. A local bank, engaged as trustee for the plan...
-
what surprised you about the future trends and supply of healthcare workers? Why might the local supply of healthcare professionals look very different than the state or national supply? Which types...
-
On January 2 of the current year, Black acquired a 5 0 % interest in New Partnership by contributing property with an adjusted basis of $ 7 , 0 0 0 and a fair market value of $ 9 , 0 0 0 , subject to...
-
Explain how replication shown in these steps from figure 6.17 would differ from those in figure 9.29. DNA DNA (3 Duplication of phage components; replication of virus genetic material 2) Penetration...
-
Software Solution is family-owned business that has been in operation for more than 15 year. The board of directors is comprised of mainly family members, plus a few professionals such as an...
-
In Problem 5-3, you drew the enantiomers for a number of chiral compounds. Now go back and designate each asymmetric carbon atom as either (R) or (S). In problem 5.3 (a) (b) (c) (d)...
-
A solution of 2.0 g of (+)-glyceraldehyde, HOCH2-CHOH-CHO, in 10.0 mL of water was placed in a 100-mm cell. Using the sodium D line, a rotation of +1.74o was found at 25 C. Determine the specific...
-
A solution of 0.50 g of (-) - epinephrine (see Figure 5-15) dissolved in 10.0 mL of dilute aqueous HCl was placed in a 20-cm polarimeter tube. Using the sodium D line, the rotation was found to be...
-
In 2022, Anne had a $24,000 net short-term capital loss and deducted $3,000 as a capital loss deduction. In 2023, Anne has a $36,000 0%/15%/20% long-term capital gain and no other capital gain or...
-
There is little evidence to suggest that ABC systems improve return on assets in entities that use it. True False
-
A kennel designed for boarding small dogs has current assets of $8,000, current liabilities of $2,000, and fixed assets of $25,000. The small company has a current ratio of:Group of answer...

Study smarter with the SolutionInn App


