Noting the LX character of the allyl ligand in Table 18.1, sketch the allylmetal interaction, showing both
Question:
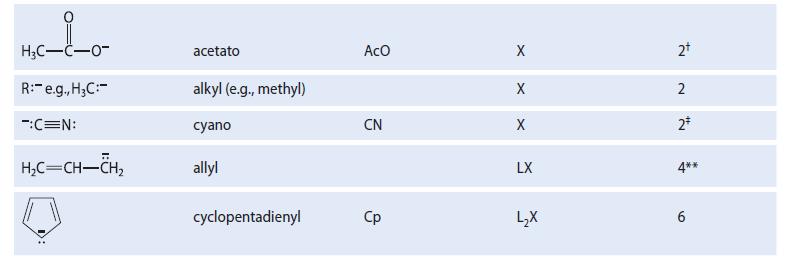
Noting the LX character of the allyl ligand in Table 18.1, sketch the allyl–metal interaction, showing both L-type and X-type bonds. Use M as a general metal.
Transcribed Image Text:
TABLE 18.1 Some Typical Ligands Used in Transition-Metal Chemistry Abbreviation Ligand H₂N: H₂O: R3P: (R = alkyl, aryl) :c=0: H₁₂C=CH₂5 CH₂C=N: F, Cl, Br, I H™ Name ammine aquo trialkylphosphino, triarylphosphino carbonyl ethylene acetonitrile benzene halo (e.g., chloro) hydrido со MeCN X Type L L L L L L L3 X X Electron count* 2 2+ 2 2* 2 295 6 2+ 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The bond to one allylic carbon is an Xtype bond That is if we break the me...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Emily Jackson (Social Security number 765-12-4326) and James Stewart (Social Security number 466-74-9932) are partners in a partnership that owns and operates a barber shop. The partnership's first...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Modify Lookup to make a program LookupAndPut that allows put operations to be specified on standard input. Use the convention that a plus sign indicates that the next two strings typed are the...
-
(a) In the reference frame of the muon in Problem 4, how far does the laboratory travel in a typical lifetime of 2 s? (b) What is this distance in the laboratorys frame?
-
Find the required outside diameter d for a steel pipe column (see figure) of length L = 11.5 ft that is pinned at both ends and must support an axial load P = 80 k. Assume that the wall thickness t...
-
Why does an accurate calculation of EVA or profitability mean that the balance sheet will have to be restated?
-
Chilczuk, S.A., of Gdansk, Poland, is a major producer of classic Polish sausage. The company uses a standard cost system to help control costs. Manufacturing overhead is applied to production on the...
-
Two artillery bases are located at the points K and M represented in the figure. Each of the bases locates an enemy airplane at point L and measures the angles of elevation to the airplane. Given the...
-
Which of the two compounds in each of the following sets should react more rapidly in a nucleophilic aromatic substitution reaction with CH 3 O in CH 3 OH? Explain your answers. (a) NO or F NO (b) NO...
-
Within each series, arrange the compounds according to increasing rates of their reactions by the S N 1E1 mechanism. Explain your reasoning. (b) Br -C=CH, A Cl -CH-CH3 A Br | -CH-CH3 B B Br C Cl...
-
The file Property Taxes contains the property taxes per capita for the 50 states and the District of Columbia. Decide whether the data appear to be approximately normally distributed by a. Comparing...
-
On January 1, 2022, Hum Company purchased 10,000 ordinary shares of Yippy Company, P100 par for P750,000. On March 10, it received 20% stock dividend. The company exchanged 4,000 ordinary shares of...
-
Cool Sips Company traded in a manual ideal machine for an automated ideal machine an gave $4,000 cash. The old machine.cost $46,500 and had a net book value of $35,500. Th old machine had a fair...
-
Complete the table given below 14.24 Complete the available-to-promise table below Period 1 Forecast 50 CO 56 257 3 4 5 6 50 50 50 50 50 17 75 50 16 14 MPS 100 100 100 ATP ? ? ?
-
The following data pertain to XYZ Company. Net income is assumed to be 8% of sales and the dividend payout ratio is 40%. The company expects its sales to increase its sales from 400,000 in 20X1 to...
-
1.3 Write a matlab program When a resistor (R), capacitor (C) and battery (V) are connected in series, a charge Q builds up on the capacitor according to the formula Q(t) = CV (1-e-t/RC) if there is...
-
A supermarket/drugstore assignment: Look at the labels of 10 different products used to control microbes, and make a list of their active ingredients, their suggested uses, and information on...
-
If there is an unrealized holding gain on available-for-sale investments, it is reported as?
-
(a) Using bond-dissociation enthalpies from Table 4-2 (page 143), calculate the heat of reaction for each step in the free-radical bromination of methane. (b) Calculate the overall heat of reaction....
-
The reaction of tert-butyl chloride with methanol Is found to follow the rate equation Rate = kf [(CH3)3C-Cl] (a) What is the kinetic order with respect to tert-butyl chloride? (b) What is the...
-
Under certain conditions, the bromination of cyclohexene follows an unusual rate law: Rate = kr [cyclo-hexene][Br2]2 (a) What is the kinetic order with respect to cyclo-hexene? (b) What is the...
-
If the court finds that a requesting spouse IS eligible for an award of spousal maintenance the court must then do which of the following: Question 24 options: Give the requesting spouse as much...
-
1)The company's additional loan for P1,500,000 was approved and release by the bank on 8/31/2024. The loan will be paid in eighteen equal monthly installments. The bank erroneously credited the...
-
The organization chart of Desjardins Group, one of the best known credit unions in the world, groups the services it offers to customers. These groupings include Insurance, Securities, Personal...

Study smarter with the SolutionInn App


