Propose a curved-arrow mechanism for each of the reactions given in Fig. P17.46. (b) CH3(CH)3 C=C-CH-Br +
Question:
Propose a curved-arrow mechanism for each of the reactions given in Fig. P17.46.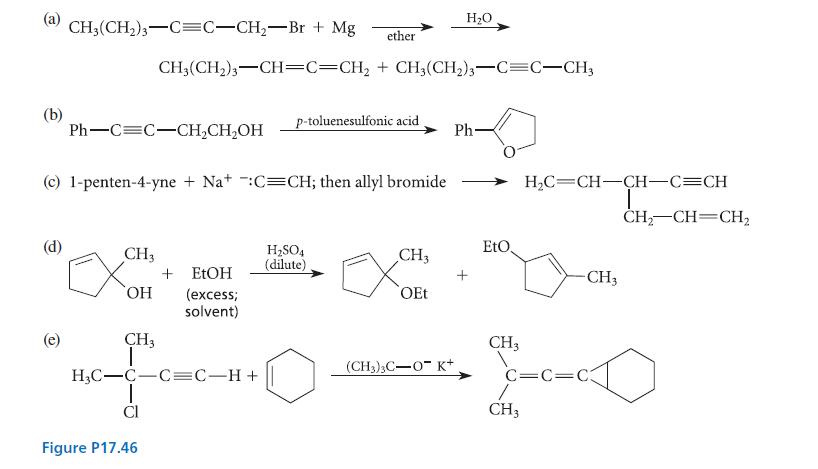
Transcribed Image Text:
(b) CH3(CH₂)3 C=C-CH₂-Br + Mg (e) Ph-C=C-CH₂CH₂OH CH3 (c) 1-penten-4-yne + Na+ :C=CH; then allyl bromide + EtOH OH (excess; solvent) CH3 CH₂(CH₂)3-CH=C=CH₂ + CH3(CH2)3-C=C-CH3 H₂C-C-C=C-H+ Cl Figure P17.46 ether p-toluenesulfonic acid. H₂SO4 (dilute) CH3 OEt H₂O (CH3)3C-OK+ Ph- EtO. CH3 H₂C CH-CH-C=CH L CH3 -CH3 C=C=C3 CH₂ CH CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Propargylic Grignard reagents like allylic Grignard reagents are an equilibrium mixture of two con...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The total amount paid in by__________for the shares they purchase is described as common stock. Select answer from the options below creditors bondholders employees stockholders
-
Give a curved-arrow mechanism for each of the reactions given in Fig. P20.51. OC,H, dil. HCI (catal ,- OC2Hs an orthoester 0 CIH O C CH dil HOI (catalyst) CH OH (e) H,C ,--, carbon monoxide CH Ph Ph...
-
Give a curved-arrow mechanism for each of the reactions given in Fig. P20.52. (a) (b) OEt 1 H3C-C-OEt + HO T OEt an orthoester (c) HC H3C C-CH3 COH dil. HCl (catalyst) CH3OH + C=CH + :C=O: dil. HCI...
-
Modify BST to add a method rangeSearch () that takes two keys as arguments and returns an iterable over all keys that are between the two given keys. The running time should be proportional to the...
-
An interstellar spaceship travels from the earth to a distant star system 12 light-years away (as measured in the earths frame). The trip takes 15 years as measured on the ship. (a) What is the speed...
-
A spherical steel pressure vessel (diameter 480 mm, thickness 8.0 mm) is coated with brittle lacquer that cracks when the strain reaches 150 ( 10-6 (see figure). What internal pressure p will cause...
-
An experiment was recently carried out where a child, an astrologer and a financial analyst were each given 10,000 to invest for eight years. Who do you think achieved the best results?
-
The Diagonal Stamp Company, which sells used postage stamps to collectors, advertises that its average price has increased from $1 to $5 in the last 5 years. Thus, management states, investors who...
-
find the area of the shaded (010) 3 y= x-4x (3,15) haded regiony, bl (option) Ag B D, 33 4
-
Specify the relationship(s) of the compounds in each of the following sets. Choose among the following terms: identical compounds, epimers, anomers, enantiomers, diastereomers, constitutional...
-
Consider the relative rates of the two solvolysis reactions in acetic acid solvent shown in Fig. P17.50. (a) Suggest a reason that compound A undergoes solvolysis much faster than compound B. (b)...
-
Compare the values of y and dy if y = f(x) = x 3 + x 2 2x + 1 and x changes (a) From 2 to 2.05 (b) From 2 to 2.01.
-
Please post a comment on the following? "Data analytics is becoming a very important tool, even when it comes to sports, and more and more teams are creating separate departments to do this work. By...
-
how would I do what's in the highlighted area Your supervisor has asked you to complete a two-part project. The first part involves joining ServerSA1 to the company Active Directory domain, and the...
-
Company Z is looking at potentially introducing a new product, and has compiled the following annual estimates: Revenue: $ 3 0 , 0 0 0 Fixed Costs: $ 1 8 , 0 0 0 ...
-
1. Recall the "falling chain" problem from class and assume the chain to have length X and mass m. Y 9 In terms of the distance y, the chain's kinetic energy and potential energy are given by m 47 (A...
-
The following table shows the crude steel production in the USA, in millions of metric tons. Year Crude Steel Production 1980 0 80 1988 8 92 91 1991 11 (A) Find a quadratic function f(x) = ax2 + bx +...
-
Explain this statement: The prevalence of gonorrhea is higher in women, but the incidence of gonorrhea is higher in men.
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
Without drawing the MOs, state whether the 7r-molecular orbital 6 in 1,3,5,7,9-decapentaene (a 10-carbon conjugated alkene) is symmetric or anti symmetric with respect to the reference plane; is...
-
What do the pericyclic selection rules have to say about the position of equilibrium in each of the reactions given in Fig. P27.30? Which side of each equilibrium is favored and why? Fig. P27.30 (a)...
-
What stereoisomer of A also gives compound C on heating?
-
Tags in QBO can be added to which of the following? Multiple Choice Invoices Expenses Bills All of the choices are correct
-
Which statement is true regarding e-prescribing (eRx)? a. eRx is available in all EHR systems. b. CPOE replaces eRx in a certified EHR system. c. eRx is not required for ARRA incentive payments is...
-
Concerning the credit for income taxes paid to another state, which of the following statements is true? Question 11Select one: a. An Oregon resident is allowed a credit for taxes paid to another...

Study smarter with the SolutionInn App


