Provide two different reaction sequences that could be used to synthesize 4-methoxy-3-methylbiphenyl. Both sequences, however, should start
Question:
Provide two different reaction sequences that could be used to synthesize 4-methoxy-3´-methylbiphenyl. Both sequences, however, should start with both p-bromoanisole and m-bromotoluene.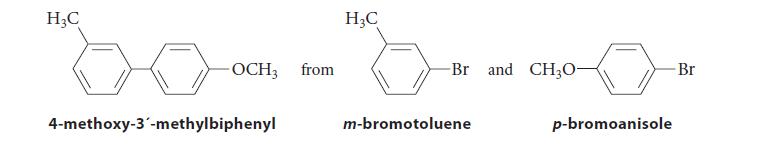
Transcribed Image Text:
H₂C -OCH3 from 4-methoxy-3'-methylbiphenyl H₂C -Br and CH3O- m-bromotoluene p-bromoanisole Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The starting materials for a Suzuki coupling can be determined by mentally splitting the arylar...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
In 2015, John decided to start up his own brewery, Tasmania Microbrewery Inc (TMI). His family supported his decision and joined him in investing in the business. TMI began operations on January 1,...
-
In 2009, David Stott quit his job at a large beer company to start his own brewery, Arizona Microbrewery, Inc. (AMI). His family supported his decision and invested in the business along with David....
-
Use a software package such as Matlab or Mathematica to program the example described in section 7.3. (a) Assume the environmental regulator ignores the impact on the labor market and sets an...
-
A spaceship of proper length L = 400 m moves past a transmitting station at a speed of 0.76c. At the instant that the nose of the ship passes the transmitter, clocks at the transmitter and in the...
-
A three-span continuous beam ABCD with three equal spans supports a uniform load of intensity q (see figure). Determine all reactions of this beam and drawn the shear-force and bending-moment...
-
In practice, what is the most serious problem raised by real options?
-
Taos Company's record of transactions concerning part X for the month of April was as follows. Instructions (a) Compute the inventory at April 30 on each of the following bases. Assume that perpetual...
-
Pena Company is considering an investment of $27,000 that provides net cash flows of $9,000 annually for four years. (0) If Pena Company requires a 10% return on its investments, what is the net...
-
Draw a curved-arrow mechanism for the last (acid-catalyzed hydrolysis) step of Eq. 18.50. MgBr OCH3 + B(OCH3)3 B(OCH3)3 MgBr OCH3 H0 HO B(OH)2 OCH3 + 3 CH3OH 2+ + Mg+ + Br (18.50)
-
What product is expected when cyclopentene reacts with iodobenzene in the presence of triethylamine and a Pd(0) catalyst?
-
Werner Chemical, Inc., leased a protein analyzer on September 30, 2021. The five-year lease agreement calls for Werner to make quarterly lease payments of $391,548, payable each September 30,...
-
The experimental protocol makes it possible to understand why you must carry out an experiment, the objectives of your experiment, the hypotheses tested, the outline of the course of the experiment...
-
Water in a piston-cylinder assembly undergoes a constant-pressure process at 30 bar from T = 267.9C to saturated vapor. Determine the work for the process, in kJ per kg of water.
-
Just a few hints that will hopefully help you do y0ur best work in completing competency one Since this is a practical course, the expectation is that you will create the most realistic analysis...
-
James and Brittany are married, and they are filing their 2020 federal income taxes jointly. Suppose $80 is deducted from each of James's monthly paychecks for health insurance, and $125 is deducted...
-
You have $25,000 in an investment account today. How much will be in the account in 30 years if the account earns (a) 8% per year, (b) 8% compounded semiannually, (c) 8% compounded quarterly, (d) 8%...
-
If the following patient specimens produced positive cultures when inoculated and grown on appropriate media, indicate whether this result indicates a disease state and why or why not: Urine Lung...
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
(a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane. (b) Explain why free-radical halogenations usually gives mixtures of products. (c) How could...
-
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
Camby Corp. can make one of the following four products: Product 1 Product 2 Product 3 Product 4 Selling price per unit $94.00 $211.00 $291.00 $42.00 Variable cost per unit $35.00 $117.00 $207.00...
-
Gastow Pumps is a manufacturer of commercial and heavy industrial Pumps. The firm's two product lines are called Directlift and Gravity. The primary raw materials are flexible steel sheets, and 23cm...
-
Caso de negocio: RoboTech. Irrumpiendo en el mercado estadounidense Con la informacin que Chen tena disponible en 2009, crees que la decisin de invertir 45 millones de dlares para diversificar...

Study smarter with the SolutionInn App


