The following compound, unlike most phenols, is soluble in neutral aqueous solution, but insoluble in aqueous base.
Question:
The following compound, unlike most phenols, is soluble in neutral aqueous solution, but insoluble in aqueous base. Explain this unusual behavior.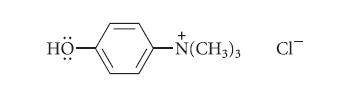
Transcribed Image Text:
HỌ -N(CH3)3 CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The solubility of most phenols in base due to their 1 charge when ionized Their c...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Toronto Tutors has been offering home tutoring services around the Toronto area since 2008. Roberta Draper started the business from her home, setting up tutoring sessions for math, science, and...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
Consider again examples 9.2 and 9.3 , and once again assume that there are two states of the world for each random variable, denoted by V L j , V H j and n L , n H . Denote the probabilities for...
-
The time interval between the reception of the flashes at A and B in Problems 26 and 27 is 75 min according to the observer in S. How much time does he expect to have elapsed on the clock at A during...
-
A propped cantilever beam AB of length L, and with guided support at A, supports a uniform load of intensity q (see figure). (a) Evaluate the strain energy of the beam from the bending moment in the...
-
Is the Heineken share more or less risky than the whole of the market? Why?
-
After the accounts have been adjusted at April 30, the end of the fiscal year, the following balances were taken from the ledger of Nuclear Landscaping Co.: Felix Godwin, Capital......................
-
dx.y) Solve the system u=2x-y, v=2x+2y for x and y in terms of u and v. Then find the value of the Jacobian (u,v) The function for x in terms of u and v is x- The function for y in terms of u and v...
-
Given the structure of phenanthrene, draw structures of (a) 9,10-phenanthroquinone (b) 1,4-phenanthroquinone phenanthrene L 5 00 a N 10
-
Which of the two phenols in each set is more acidic? Explain. (a) 2,5-dinitrophenol or 2,4-dinitrophenol (b) phenol or m-chlorophenol (c) OH CH 0 or OH CH=0
-
With regard to interest charged on partners drawings which one or more of the following statements is/are correct? (a) Interest is charged to ensure partners do not draw their profit share at all (b)...
-
Machinery was purchased for $440000 on January 1, 2022. Freight charges amounted to $15000 and there was a cost of $40000 for building a foundation and installing the machinery. It is estimated that...
-
In the context of business negotiation, what communication techniques are most effective for achieving favorable outcomes? Discuss the balance between assertiveness and collaboration, and the role of...
-
A foreign investor is interested in entering an economic sector of the Dominican Republic, for this he selected 5 sectors and thus evaluate their behavior. This was based on data from the Central...
-
A pioneering e-commerce platform is helping women in East Africa access menstrual care, personal care and women's health products. The platform is optimised to provide confidential access to products...
-
Complete the income portion of Forms 1120 and 1125-A for the corporation listed below. The Computer Executive corporation presents the following information in its books: Income received from sales...
-
Shigella, Mycobacterium, and numerous other pathogens have developed mechanisms that prevent them from being killed by phagocytes. a. Suggest two or three factors that help them avoid destruction by...
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Each of the following descriptions applies to more than one alkane. In each case, draw and name two structures that match the description. (a) An isopropylheptane (b) A diethyldecane (c) A...
-
Give the IUPAC names of the following alkanes. (a) CH3C(CH3)2CH(CH)CH3)CH2CH2CH(CH3)2 (b) (c) (d) (e) (f) (g) (h) CH,CH CHCH CH, CH CH CH,CHCH CH,CHCH CH,CH CH,CH, CH, CH,CH, CH,CH,CH, C(CH,CH),...
-
Construct a graph, similar to Figure 3-11, of the torsional energy of 3-methylpentane along the C2-C3 bond. Place C2 in front, represented by three bonds coming together in a Y shape, and C3 in back,...
-
Gencon Contracting has a blanket contractors equipment policy. If Gencon acquires some new equipment, what kind of coverage is probably provided on this equipment? (Search Chapter 9) a. No coverage...
-
rewrite "I believe that my high standard of ethical conduct is shown in my assignment that I have submitted. I give credit where credit is due, and if I have questions concerning APA guidelines, I...
-
In December 2022, Surf Ltd received a claim from one of its customers commencing a lawsuit alleging faults in the construction of a bridge in a housing estate. This lawsuit amounts to a total of $2.5...

Study smarter with the SolutionInn App


