The following molecule has a barrel shape (in which the benzene rings are the walls of the
Question:
The following molecule has a barrel shape (in which the benzene rings are the “walls” of the barrel). It forms a noncovalent complex with the iodide salt of acetylcholine in chloroform solvent.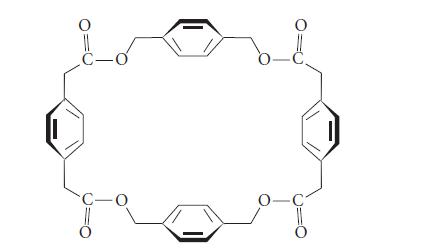
Describe the orientation of the acetylcholine molecule within the complex.
Transcribed Image Text:
O C-o C-O 8 O -C₂ 0-C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The positive end of the acetylcholine ion would orient its...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The N-methylquinolinium ion forms a noncovalent complex with molecule A in water that has a standard free energy of dissociation G d 5 28.9 kJ mol 1 (6.9 kcal mol 1 ). The neutral molecule...
-
Identify the process evaluation article that you chose and explain why you selected this example. Describe the purpose of the evaluation, the informants, the questions asked, and the results of the...
-
Which choice is greener in a chemical process? Explain. (a) Benzene as a solvent or water as a solvent. (b) The reaction temperature is 500 K, or 1000 K. (c) Sodium chloride as a by-product or...
-
What are some Marketing Strategies for Delivering Objectives Polestar (Car company) has used or using?
-
What is the maximum penalty and prison term that can be charged to a CEO and/or CFO under the Sarbanes-Oxley Act?
-
What does the rate of return on common stock show, and how is it calculated?
-
Journalize the following transactions of Cramer, Inc., which ends its accounting year on June 30: Apr 1 Jun 6 30 Loaned $20,000 cash to R. Simpson on a one-year, 8% note. Sold goods to Friday, Corp.,...
-
Following are Nintendo's revenue and expense accounts for a recent March 31 fiscal year-end (yen in millions). Prepare the company's closing entries for its revenues and its expenses. Net sales...
-
On Jan 1, 2019, AML Company issued bonds. The compounding period is annually. AML reported the following items (all the items are related to this bond). From the balance sheet: 1-Jan-19 31-Dec-19...
-
Using the Hckel 4n + 2 rule, determine whether each of the following compounds is likely to be aromatic. Explain how you arrived at the -electron count in each case. (a) (c) CO (b) -C=C- (d) :1
-
Which of the compounds or ions in Problem 15.38 are likely to be antiaromatic? Explain. Problem 15.38 Which of the following species should be aromatic by the Hckel 4n + 2 rule? (a) thiophene G O H H...
-
An O2 adsorption isotherm was obtained at 443 K for a 2.43% Ag/SiO2 catalyst and the uptake results are given below. No irreversible adsorption occurs on the silica. What is the dispersion of the...
-
(a) Why do some businesses keep a petty cash book as well as a cash book? (b) Kathryn Rochford keeps her petty cash book on the imprest system, the imprest being 25. For the month of April 2017 her...
-
What individual in your life do you need to see differently?
-
Write a recursive method to multiply two positive integers using repeated addition.
-
Write a complete program to compare the temperatures of three different cities and find the hottest city. First, implement a class called Thermometer as described below: A. Thermometer has one...
-
Of course, if the square of the velocity is a constant in the curved fourdimensional space, this is not the case if one visualizes the phenomenon in a Euclidean plane. Consider the case of free...
-
Overnight Laundry is considering the purchase of a new pressing machine that would cost $100,000 and would produce incremental cash flows of $25,000 annually for 6 years. The machine has a terminal...
-
Suppose you need to answer any four of seven essay questions on a history test and you can answer them in any order. a. How many different question combinations are possible? b. What is the...
-
Propose structures for compounds that meet the following descriptions: (a) C5H8, with IR absorptions at 3300 and 2150 cm1 (b) C4H80, with a strong IR absorption at 3400 cm1 (c) C4H80, with a strong...
-
How could you use infrared spectroscopy to distinguish between the following pairs of isomers? (a) HC CCH2NH2 and CH3CH2C N (b) CH3COCH3 and CH3CH2CHO
-
Two infrared spectra are shown. One is the spectrum of cyclohexane, and the other is the spectrum of cyclohexane. Identify them, and explain youranswer. (a) 100 80 60 20 - 1000 4000 3500 3000 2500...
-
Should People Be Paid To Stay Healthy? After reading the article, Should People Be Paid To Stay Healthy? , which side do you stand on this issue? Is it ethical for businesses to reward or penalize...
-
Swiss Banks, like UBS and Credit Suisse, have a culture ofsecrecy for their depositors, and this culture has been supportedby Swiss government policy. What problems has this policy causedfor the...
-
Blue Ridge Hiking Company has an estimated sales price of theheadlamp is RM19.99 per unit. The variable cost per unit isRM10.29. Blue Ridge Hiking Company has annual fixed costs totallingRM109,500....

Study smarter with the SolutionInn App


