The reaction given in Fig. P18.74 occurs readily at 95C (X = halogen). The relative rates of
Question:
The reaction given in Fig. P18.74 occurs readily at 95°C (X = halogen). The relative rates of the reaction for the various halogens are 290 (X = F), 1.4 (X = Cl), and 1.0 (X = Br). When a nitro group is in the para position of each benzene ring, the reaction is substantially accelerated. Give a detailed mechanism for this reaction, and explain how it is consistent with the experimental facts.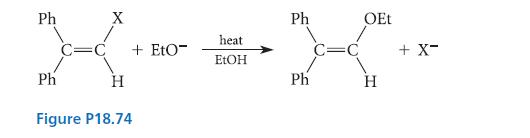
Transcribed Image Text:
Ph X X Ph H C=C + Eto- Figure P18.74 heat EtOH Ph Ph C=C OEt H + X-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Because vinylic halides do not undergo Sy2 or Syl reactions this s...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
This data table contains the listed prices and weights of the diamonds in 48 rings offered for sale in The Singapore Times. The prices are in Singapore dollars, with the weights in carats. Use price...
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P27.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
This unit addresses the role of the technology professional in an organization and how information technology (IT) systems influence organizational strategies. This case study analyzes two retailers'...
-
Write a filter that reads text from an input stream and prints it to an output stream, removing any lines that consist only of whitespace.
-
A light beam moves along the y axis with speed c in frame S, which is moving to the right with speed V relative to frame S. (a) Find the x and y components of the velocity of the light beam in frame...
-
Find the horizontal deflection δh and vertical deflection δn at the free end C of the frame ABC shown in the figure. (The flexural rigidity EI is constant throughout the...
-
You receive BC100,000 which you decide to save for your old age. You are now 20. What sort of investment should you go for? Perform the same analysis as if it happened when you are 55 and 80.
-
On January 1, 2010, Crouser Company sold land to Chad Company, accepting a two-year, $150,000 non-interest-bearing note due January 1, 2012. The fair value of the land was $123,966.90 on the date of...
-
21 Evaluate the iterated integral [18x2y5dy dx. | -1 0 2 1 SS18x2y5 dy dx = (Simplify your answer.)
-
Give the structure for each of the following compounds. (a) Isobutryaldehyde (b) Valerophenone (c) O-bromoacetophenone (d) G-chlorobutyraldehyde (e) 3-hydroxy-2-butanone (f)...
-
Predict the product of the Stille reaction between ethynyltrimethylstannane, HC CSn(CH 3 ) 3 , and phenyl triflate, PhOTf, in the presence of Pd(PPh 3 ) 4 and excess LiCl.
-
What type of layout(s) would be appropriate for: a. A grocery store? b. Home construction? c. Electronics assembly? d. A university?
-
S Bill's Wrecker Service has just completed a minor repair on a tow truck. The repair cost was $940, and the book value prior to the repair was $4,810. In addition, the company spent $6,500 to...
-
7) Among the given 207 Pb NMR spectra, the correct spectrum due to tetramethyllead is (Given: 2 J207 Pb-H = 60Hz) L l O
-
On January 1 , 2 0 2 0 , Larkspur Company issued bonds of face value $ 2 8 0 , 0 0 0 ( 2 8 0 bonds of $ 1 , 0 0 0 face value ) , with 8 % coupon. Similar bonds trade at a yield of 1 2 % in the...
-
X = F D E 3 = 5 7 $ 3 0 1 2 3 4 3 6 7 13 19 0 1 2 3 4 5 6 a 18. 3 Month September October November December The Data Table and Aggregate Demand Forecast below provide information related to one of...
-
"What are the key aspects of the recruitment and selection function, as well as the training and development function within organizations?"
-
A recent study by the CDC reported that only 73% of children in the United States are being adequately vaccinated. Several million children are at high risk for infections. a. Name some factors that...
-
During the year land was revalued and the surplus reported as Revaluation surplus; and an asset costing 80,000, written down to 38,000, was sold for 40,000. Identify the cost of any non-current...
-
N-Methylpyrrolidine has a boiling point of 81 °C, and piperidine has a boiling point of 106 °C. (a) Explain this large difference (25 °C) in boiling point for these two isomers. (b)...
-
Predict the hybridization, geometry, and bond angles for the carbon and nitrogen atoms in acetonitrile (CH3C N:).
-
Which of the following pure compounds can form hydrogen bonds? Which can form hydrogen bonds with water? Which ones do you expect to be soluble in water? (a) (CH3CH2)2NH (b) (CH3CH2)3N (c)...
-
Companies are expected to have the strongest internal controls over which the following types of data: Fixed asset records customer information bank account deposit and balance information...
-
Which of the following is not a general rule for allocating nonbusiness income? Multiple Choice Rental income for investment property to state of commercial domicile Interest and dividends to the...
-
Brothers Inc. expects earnings of $8,000,000.00 in the current year on 6,000,000 outstanding shares of common stock. The company is considering the effects on reported earnings of issuing an...

Study smarter with the SolutionInn App


