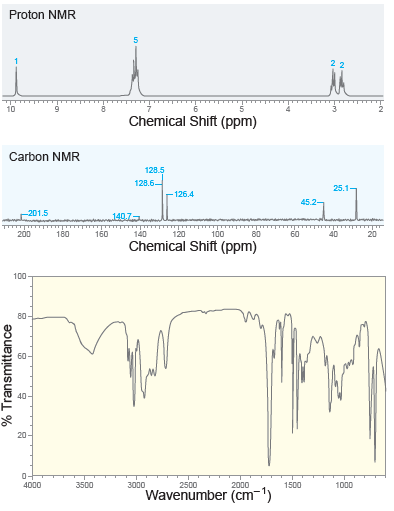
A compound with molecular formula C 9 H 10 O exhibits the following spectra ( 1 H
Question:
Transcribed Image Text:
Proton NMR 10 Chemical Shift (ppm) Carbon NMR 128.5 128.6- 25.1- 126.4 201.5 140.7 160 140 120 100 200 180 80 60 40 20 Chemical Shift (ppm) 100 80 - 60- 40- 20- 0- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-') % Transmittance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 9 H 10 O exhibits a strong signal at 1687 cm 1 in its IR spectrum. The 1 H and 13 C NMR spectra for this compound are shown below. Identify the structure of this...
-
Identify the compound with molecular formula C8H10O that gives the IR and 1H NMR spectra shown in Figure 14.23. 23 16 27 2 29 35 13 14 15 16 800 200 2400 200 0 6 (ppm) frequency
-
Propose a structure for a compound with molecular formula C 3 H 8 O that exhibits the following 1 H NMR and 13C NMR spectra: Proton NMR 0.5 5.0 4.5 4.0 3.5 3.0 2.0 1.5 1.0 25 Chemical shift (ppm)...
-
Every real number is either a/an number or a/an_______ number.
-
Explain the differences among the three major forms of bankruptcy: Chapter 7, Chapter 11, and Chapter 13.
-
A set of objects is given, together with operations of addition and scalar multiplication. Determine which sets are complex vector spaces under the given operations. For those that are not, list all...
-
Identify the costs of renting.
-
A used-car broker needs to transport his inventory of cars from locations 1 and 2 in Figure to used-car auctions being held at locations 4 and 5. The costs of transporting cars along each of the...
-
. Question 6 Smart Designs' inventory (for one item) activity for April is shown below. Smart Designs use a perpetual inventory system: Date Transaction Units Unit cost 1 April Beginning inventory...
-
An aqueous solution containing 35 wt% MgSO 4 is fed to an evaporative crystallizer operating at 50F. The vapor generated is 20% by mass of the feed. The solution, which contains 23 wt% MgSO 4 , and...
-
Solid methanol in thermal contact with the surroundings is reversibly melted at the normal melting point at a pressure of 1 atm. Are S, S surroundings , and S total positive, negative, or zero?...
-
Can incandescent lighting be regarded as an example of cogeneration during the heating season? In a season where air conditioning is required?
-
Classify each of the following aqueous solutions as a nonelectrolyte, weak electrolyte, or strong electrolyte: (a) LiClO4 (b) HClO (c) CH3CH2CH2OH (propanol) (d) HClO3 (e) CuSO4 (f) C12H22O11...
-
c) The figure below shows the federal funds rate (interbank rate in the US) and the interest on reserves from 2014-2018. How does this figure show the usefulness of interest on excess reserves as a...
-
Create a script that loops through all the stocks for one year and outputs the following information: The ticker symbol Yearly change from the opening price at the beginning of a given year to the...
-
24 Let A = , find A50. 12
-
Millie Industries manufactures two products, Monogram and Signature, for industrial use. Millie's manufacturing facility is highly automated, with very little direct labour being involved in the...
-
An ordinary share that pays half - yearly dividends is due to make a dividend payment of 5 . 8 0 per share in exactly 4 months time. Thereafter, future dividends are assumed to increase at a compound...
-
Find the area of the shaded region. The graphs depict IQ scores of adults, and those scores are normally distributed with a mean of 100 and a standard deviation of 15 (as on the Wechsler test). 1 20
-
Aztec Furnishings makes hand-crafted furniture for sale in its retail stores. The furniture maker has recently installed a new assembly process, including a new sander and polisher. With this new...
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
We can conceive of a stepwise version of the SN2 reaction consisting of a Lawis acid-base dissociation followed by a Lewis acid-base association. (Nuc:- = a nucleophile.) (a) Why should the stepwise...
-
We can conceive of a stepwise version of the SN2 reaction consisting of a Lawis acid-base dissociation followed by a Lewis acid-base association. (Nuc:- = a nucleophile.) (a) Why should the stepwise...
-
Understanding the external business environment requires marketers to be aware of: O changes in the law which impact how business is conducted O their customers' wants and needs O how employees are...
-
Data Warehouses are primarily used to manage the current and timely operational data. True False SUBMIT
-
A customer's personal philosophy is an influence on his/her adequate service expectations. True False

Study smarter with the SolutionInn App


