Cinchocaine is a long-acting local anesthetic used in spinal anesthesia. Identify the most basic nitrogen atom in
Question:
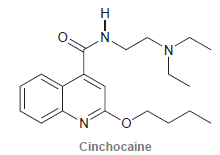
Transcribed Image Text:
Н N. N. Cinchocaine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
I...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structure of lysergic acid diethylamide (LSD), a potent hallucinogen containing three nitrogen atoms. One of these three nitrogen atoms is significantly more basic than the other two....
-
Which is the most basic nitrogen in each compound. Explain your choices. (a) (b) (c) NH2 HN
-
Novocaine, used as a local anesthetic by dentists, is a weak base (Kb = 8.91 10-6). What is the ratio of the concentration of the base to that of its acid in the blood plasma (pH 5 7.40) of a...
-
The annual growth rate in EPS is 3%. The P/E ratio is 12x. EPS last year was $1.00. Calculate What should the stock price be in 4 years?
-
"The information below relates to Milton Company's trading securities in 2012 and 2013. (a) Prepare the journal entries for the following transactions. January 1, 2012 Purchased $300,000 par value of...
-
List the four important features of 1H NMR and the information you can derive from them.
-
Ethos Day School is a private elementary school. In addition to regular classes, after-school care is provided between 3:00 p.m. and 6:00 p.m. at SF12 per child per hour. Financial results for the...
-
A CBS News/new York times survey found that 97% of Americans believe that texting while driving should be outlawed (CBS News website, January 5, 2015). a. For a sample of 10 Americans, what is the...
-
7. Instantaneous velocity Consider the position function s(t) = -1612+1281 (Exercise 13). Complete the following table with the appropriate average velocities. Then make a conjecture about the value...
-
The hydroelectric plant has a catchment area of 120 square km. The available runoff is 50% with an annual rainfall of 100 cm. Ahead of 250 m is available on an average. The efficiency of the power...
-
What is the rule of three? Suppose you worked for a small firm in a large industrysuch as a small manufacturer of furniture. How might you actually use the rule of three to enhance your firms...
-
How does the BCG matrix help marketers decide which products to offer? According to the matrix, which types of products are most desirable, and why?
-
Select a particular managerial job and observe the manager in action, preferably over an extended period of time. a. Make notes, with supporting examples, of the proportion of the manager's work and...
-
What is the IRR of an investment of \($1,000\) that yields cash flows of \($600\) in each of the first two years?
-
Your grandfather put $1,000 in the bank for you 48 years ago. The bank paid 6 percent interest compounded annually during this time period. How much is your grandfathers deposit worth today?
-
What does redundant hardware mean? Is this an unnecessary cost to a business?
-
What is the break-even price when a company expects to sell 5,000 units, the fixed costs are \($50,000,\) and the variable costs are \($30\) per unit?
-
Alpha makes a product with a fixed cost of \($400,000\) and a variable cost of \($100\) per unit. The sales price is \($200\) per unit. What is the break-even point for the product? How many units...
-
What are different ways investigators can obtain hard-to-get documentary evidence?
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
Show how the product of part (a) can be converted into estrone. Me O HIH HO estrone
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P27.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
(a) What type of pericyclic reaction is required to form benzene from Dewar benzene? (b) Explain why Dewar benzene, although a very un-stable molecule, is not readily transformed to benzene....
-
Good afternoon, is my assignment instructions and required columns/rows of data. This 'ask your question' box will not allow me to attach the actual Excel file that has my formulas and calculations,...
-
Con los conocimientos adquiridos en la unidad y las orientaciones del facilitador: Evala la eficiencia del proceso de produccin en una empresa de manufacturera y refiere, si este se apega a lo...
-
Clothing Company has two service departments purchasing and maintenance, and two production departments fabrication and assembly. The distribution of each service department's efforts to the other...

Study smarter with the SolutionInn App


