Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite
Question:
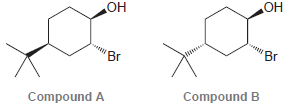
Transcribed Image Text:
OH OH 'Br 'Br Compound B Compound A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
T his process for epoxide formation involves deprotonation of the hyd...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following two compounds. Monochlorination of one of these compounds produces twice as many stereoisomeric products as the other. Draw the products in each case, and identify which...
-
Consider the following two compounds. These compounds are stereoisomers of 1,2,3-trimethylcyclohexane. One of these compounds has three chirality centers, while the other compound has only two...
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Determine whether the statement is true or false. If it is true, explain why. If it is false, explain why or give an example that disproves the statement.
-
PepsiCo ran an ad and promotional campaign in 1996 called "Drink Pepsi Get Stuff" campaign. The enormously successful campaign allowed customers to claim prizes in exchange for points on PepsiCo...
-
Large quantities of liquefied natural gas (LNG) are shipped by ocean tanker. At the unloading port provision is made for vaporization of the LNG so that it may be delivered to pipelines as gas. The...
-
The controls in a computerized system are classified as: a. input, processing, and output. b. input, processing, output, and storage. c. input, processing, output, and control. d. input, processing,...
-
Go to Extended Case 29.1, Citizens National Bank of Jessamine County v. Washington Mutual Bank, 309 S.W.3d 792 (2010), on pages 567568. Read the excerpt and answer the following questions. (a) Issue:...
-
Smith-Kline Company maintains inventory records at selling prices as well as at cost. For 2021, the records indicate the following data: ($ in 000s) Cost Retail Beginning inventory $ 79 $ 124...
-
Prepare a PowerPoint Presentation that discusses each element that you would include in a contract for the building of a home. You do NOT include the concepts that lead to contract creation. This...
-
Assume that the equation of state for a gas can be written in the form P(V m b(T)) = RT. Derive an expression for = 1/V (V /T)P and = 1/V (V /P)T for such a gas in terms of b(T), db(T)/dT, P, and...
-
One mole of Ar initially at 310. K undergoes an adiabatic expansion against a pressure P external = 0 from a volume of 8.5 L to a volume of 82.0 L. Calculate the final temperature using the ideal gas...
-
The author is considering deleting the previous sentence. If the sentence were deleted, the essay would primarily lose: F. An example of how a dog communicates with its owner. G. Support for the...
-
Teachers' Salaries The average annual salary for all U.S. teachers is $47,750. Assume that the distribution is normal and the standard deviation is $5680. Find these probabilities of the earnings of...
-
88. A certain college has g good courses and b bad courses, where g and b are positive integers. Alice, who is hoping to find a good course, randomly shops courses one at a time (without replacement)...
-
The shares of a newly listed company are doubling in value every 12 hours. If you bought $3,000 worth of the stock when it was first offered: Determine the appropriate model giving the value of your...
-
The income statement, balance sheets, and additional information for Video Phones, Incorporated, are provided. Net sales Expenses: VIDEO PHONES, INCORPORATED Income Statement For the Year Ended...
-
. Jeremy is thinking of starting up a candle manufacturing business. The initial outlay for equipment, moulds, and other required production equipment is $15,000. Working part time on this hobby...
-
Stephanie and Rosie are classmates who each conduct their own study on the same topic. Stephanie has a sample size of 150. Rosie has a sample size of 285. Which student is more likely to retain the...
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
Poly(ethylene terephthalate), or PET, is a polyester used to make soft-drink bottles. It is prepared by reaction of ethylene glycol with 1,4-benzenedicarboxylic acid (terephthalic acid). Draw the...
-
Show the mechanism of the nucleophilic addition reaction of an alcohol with an iso-cyanate to yield a urethane.
-
What product would you expect to obtain from catalytic hydrogenation of natural rubber? Would the product be syndiotactic, atactic, or isotactic?
-
Question 212: A user is filling out a digital create condition records form. The user tried entering a dollar sign ($) for the first input in the amount field, the form system would not allow it, and...
-
Es la discEs la disciplina de valor que consiste en la produccin de bienes y servicios con la mxima reduccin de costos fijos, optimizando los procesos para reducir los costos de transaccin y todos...
-
Oxford Company has limited funds available for investment and must ration the funds among four competing projects. Selected information on the four projects follows: Life of Investment Present Value...

Study smarter with the SolutionInn App


