Deduce the structure of a compound with molecular formula C 9 H 10 O 2 that produces
Question:
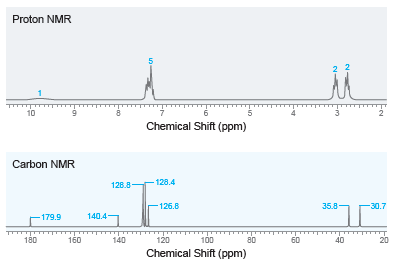
Transcribed Image Text:
Proton NMR 10 Chemical Shift (ppm) Carbon NMR - 128.4 128.8- 35.8 -126.8 -30.7 140.4 -179.9 180 160 100 140 120 80 60 40 20 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the structure of a compound with molecular formula C 5 H 12 that exhibits only one kind of proton. That is, all 12 protons are chemically equivalent.
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
Deduce the structure of a compound having the mass spectrum and 1H NMR spectrum presented in Figure 13.43.
-
Select the reasons why most professional telescopes are reflectors rather than refractors. A mirror can collect light more efficiently than a lens. Reflecting telescopes have shorter focal lengths...
-
1. According to the case, how many Yahoo!'s user accounts were stolen in 2013? 2. What types of information were stolen? 3. When did Yahoo! find out about the 1 billion user accounts breach? 4....
-
A tall pea plant with axial flowers was crossed to a dwarf plant with terminal flowers. Tall plants and axial flowers are dominant traits. The following offspring were obtained: 27 tall, axial...
-
The Stallion Division of Motortown Motors had an operating income of \($675,000\) and net assets of \($2,700,000\). Motortown Motors has a target rate of return of 23 percent. Required a. Compute the...
-
Organic Pastures is a 400-acre farm on the outskirts of the Kentucky Bluegrass, specializing in the boarding of broodmares and their foals. A recent economic downturn in the thoroughbred industry has...
-
The Cacao Butter Edibles Factory manufactures and distributes chocolate products Click the icon to view more information about Cacao Butter) Points: 0 of 10 Production and sales data for August 2020...
-
The Village of Seaside Pines prepared the following General Fund Trial Balance as of December 31, 2012, the last day of its fiscal year. a. Prepare the closing entries for December 31. (It is not...
-
Why does species richness vary from one community to another?
-
Look at Figure 1.3. Suppose that the cost of cheese falls, so that the marginal cost of producing pizza decreases. Will the MC curve shift up or down? Will the optimal amount of pizza increase or...
-
Find the function F that satisfies the following differential equations and initial conditions. F"(x) = 1, F'(0) = 3, F(0) = 4
-
CBC Bank has a small branch in Mont-Tremblant. The counter is staffed by one teller. The counter is open for five hours (300 minutes) each day (the operating capacity). It takes six minutes to serve...
-
The researcher would like to investigate whether the variables Y t and X t form a long-run relationship and if so, to what extend they bear such a relationship. Outline your research strategy, in...
-
During 2013, Rogue Corporation reported sales revenue of $610,000. Inventory at both the beginning and end of the year totaled $80,000. The inventory turnover ratio for the year was 5.0. What amount...
-
14A. If a coolant hose is deteriorating what could happen? 14B. If one radiator hose is defective and needs replacing what should be done with the other hoses? 14C. Explain the following hose...
-
Out of the 120 sample observations, if the first 110 samples were taken to regress the AR (1) model. Then, using the estimated model to predict the next 10 observations, the root mean squared error...
-
The amount of nitrogen oxides (NOX) present in the exhaust of a particular model of old car varies from car to car according to a Normal distribution with mean 1.4 grams per mile (g/mi) and standard...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Predict the products of the following reactions: (b) CH CHCCHH2CH (a) HCI H (d) CH CH2 (c) HBr CH-CH3CH2 H20 H2SO, (Addition of H20 occurs.)
-
What alkenes would you start with to prepare the following alkylhalides? CH-CH (a) (b) Br Br CI (c) (d) CH3CH2CHCH2CH2CH3
-
Show the structures of the carbocation intermediates you would expect in the following reactions: (b) CH3 (a) CH3CH2CCHCH3 HI CH H Er
-
You should be able to arrive at the audited balance by referring to the balance sheet and the adjustments, if any, that you may be able to identify based on available findings and information. If...
-
me now ROI and Investment Decisions Allard, Inc., presented two years of data for its Frozen Foods Division and its Canned Foods Division. Frozen Foods Division: Year 1 Year 2 Sales $35,200,000...
-
Question 3 Conversion costs include: I. Depreciation of the production facility II. Direct labor III. Indirect labor IV. Indirect materials A. I only OB. I, II, and III OC. I, III and IV O D. All of...

Study smarter with the SolutionInn App


