For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic,
Question:
(a)
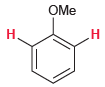
(b)
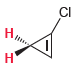
(c)
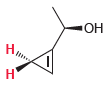
(d)
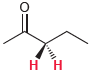
(e)
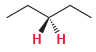
Discuss.
Transcribed Image Text:
OMe Н. Н CI H,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a Homotopic b ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
For each of the following compounds, use the nitrogen rule to determine whether the molecular weight should be even or odd. Then calculate the expected m/z value for the molecular ion. a. b. c. d. O:...
-
Engineering is a dynamic field that requires continuous learning. Discuss how you plan to acquire and apply new knowledge as needed throughout your engineering career. Address the strategies you...
-
What is intellectual property? What are three examples of intellectual properties?
-
A Drosophila embryo dies during early embryogenesis due to a recessive maternal effect allele called bicoid. The wild-type allele is designated bicoid*. What are the genotypes and phenotypes of the...
-
What is the relationship between segment reports and product reports?
-
Forecasts and actual sales of MP3 players at Just Say Music are as follows: a. Plot the data and provide insights about the time series. b. What is the forecast for November, using a two-period...
-
On January 1, 2018, Reston Company bought 25% outstanding ordinary shares of Ace Company for P1 million. The book value of the net asset acquired was P3 million. The difference was attributable to...
-
Reconsider the semiconductor speed data in Exercise 6-42. Construct a box plot of the data and write an interpretation of the plot. How does the box plot compare in interpretive value to the original...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Temperature may alternatively be defined as Prove that this definition reduces the net entropy change of two constant-volume systems filled with simple compressible substances to zero as the two...
-
3. Expand the given logarithm based on log(x), log(y) and log(z). x2z5 log
-
Suppose that the 2-year interest rates with continuous compounding in Australia and the United States are 1.6% and 2.2% per annum, respectively, and the spot exchange rate between the Australian...
-
A company produces 3 products with the following budget information available: ABC Sales GH14GH15GH18 Standard full costGH10GH10GH13 Budget Production10,000 units13,000 units9,000 units The actual...
-
NO. 8 Moana Company, produces a product called Lilo. The company buys materials needed to make a Lilo from different suppliers in Cotabato at P1.50 each. To convert the materials into Lilo, the same...
-
1. Prepare condensed divisional income statements for the year ended December 31, 2017, assuming that there were no service departmen cost allocations. Free Ride Bike Company Divisional Income...
-
The Survey of Study Habits and Attitudes (SSHA) is a psychological test that measures academic motivation and study habits. The SSHA score F of a randomly selected female student at a large...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Normally, a Trans alkene is more stable than its cis isomer Trans-Cyclooctene, however, is less stable than cis-Cyclooctene by 38.5kJ/mol. Explain.
-
Trans-Cyclooctene is less stable than cis-Cyclooctene by 38.5kJ/mol, but Trans cyclononene is less stable than cis-cyclononene by only 12.2kJ/mol. Explain.
-
Allene (1, 2-propadiene), H2C = C = CH2, has two adjacent double bonds. What kind of hybridization must the central carbon have? Sketch the bonding orbitals in allene. What shape do you predict for...
-
For each of the following taxpayers indicate the applicable filing status and the number of children who qualify for the child credit. a. Jeffrey is a widower, age 71, who receives a pension of...
-
Dividends Per Share Oceanic Company has 25,000 shares of cumulative preferred 2% stock, $50 par and 50,000 shares of $5 par common stock. The following amounts were distributed as dividends: Year 1...
-
The method that allocates costs in each cost pool using the same rate per unit is known as the Select one: a. dual-rate cost allocation method b. single-rate cost allocation method c. reciprocal cost...

Study smarter with the SolutionInn App


