For each of the following reactions determine whether ÎS for the reaction (ÎS sys ) will be
Question:
a.
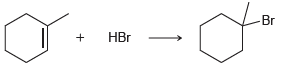
b.
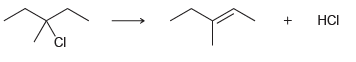
c.
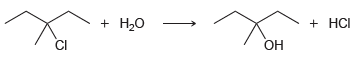
d.
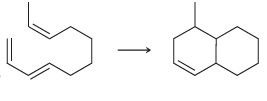
e.
Transcribed Image Text:
Br НBr HCI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
a S sys is expected to be negative a decrease in entropy because ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) For each of the following reactions, predict the sign of ÎH° and ÎS° and discuss briefly how these factors determine the magnitude of K. (b) Based on your general chemical...
-
For each of the following reactions, give the curved-arrow notation and write the analogous Bonsted acid-base reaction. H,C-CH,-Br: + :C=N: H,C-CH-C=N: + :Br:
-
For each of the following reactions. provide the following information. (a) Give the structures of all products (including stereoisomers). (b) If more than one product is formed, give the...
-
You are looking at buying a piece of real estate and you intend to borrow as much as you possibly can from a bank to buy the property. The bank you are dealing with has a requirement that the LVR for...
-
Suppose you want to perform an experiment to verify the problems that can be caused by random insert/remove pairs. Here is a strategy that is not perfectly random, but close enough. You build a tree...
-
Compare the number with the default rate in Table 18-2. Discuss what could explain the difference.
-
A ____________ links key goal-related issues with key collaboration issues to come up with general ways by which the firm will manage its affairs. (a) managerial philosophy (b) cultural symbol (c)...
-
The following financial data were reported by 3M Company for 2006 and 2007 (dollars in millions). Instructions(a) Calculate the current ratio and working capital for 3M for 2006 and 2007.(b) Suppose...
-
Discuss the working capital management of the company in respect of the recent two years. How can management address the immediate impact on liquidity? Address the following: Inventory Cash and Cash...
-
Martin's Service Station is considering entering the snowplowing business for the coming winter season. Martin can purchase either a snowplow blade attachment for the station's pick-up truck or a new...
-
Explain: The United States can make certain toys with greater productive efficiency than can China. Yet we import these toys from China. Relate your answer to the ideas of Adam Smith and David...
-
The percentage that casinos make on the average bet is called the a. Vig. b. Rip. c. Take. d. Rob.
-
A helium-filled balloon escapes a childs hand at sea level and 20.0oC. When it reaches an altitude of 3000m, where the temperature is 5.0oC and the pressure is only 0.70atm, how will its volume...
-
As the primary breadwinner in her household, Miranda is determining her life insurance needs. Her husband was injured in a work-related accident and receives social security disability (SSDI)...
-
Market data obtained from S&P Capital IQ is shown below: VMX Ltd has a beta of 1.3 and debt-to-equity ratio of 0.5 Yield on long-term Singapore government bonds = 3% Yield on AAA-rated corporate...
-
1. Explain the following types of building and construction industry contracts. Write answer in some words. Lump sum contract Cost plus contract Commercial contracts Subcontract agreements 2. What...
-
Concept testing is defined as a research method that involves asking customers questions about your concepts and ideas for a product or service before actually launching it. Thus, you can gauge your...
-
You've recently launched a computer repair company in Vevey. Your services are reasonably priced, and your value add is that you go to people's homes and are able to work on-line. Your target...
-
The Pew Internet and American Life Project asked a random sample of U.S. adults, Do you ever use Twitter or another service to share updates about yourself or to see updates about others? According...
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
Which of the following alkyl halides would you expect to undergo Friedel-Crafts reaction without rearrangement? Explain. (a) CH3CH2Cl (b) CH3CH2CH (Cl) CH3 (c) CH3CH2CH2Cl (d) (CH3) CCH2Cl (e)...
-
What is the major mono-substitution product from the Friedel-Crafts reaction of benzene with 1-chloro-2-methylpropane in the presence of AlCl3?
-
Identify the carboxylic acid chloride that might be used in a Friedel-Crafts acylation reaction to prepare each of the followingacylbenzenes: (b) (a)
-
Majestic Corporation began the year with accounts receivable, inventory, and prepaid expenses totaling $63,000. At the end of the year, Majestic had a total of $77,000 for these current assets. At...
-
Goshford Company produces a single product and has capacity to produce 155,000 units per month. Costs to produce its current sales of 124,000 units follow. The regular selling price of the product is...
-
The Town of Weston has a Water Utility Fund with the following trial balance as of July 1, 2019, the first day of the fiscal year: Cash Customer accounts receivable Allowance for uncollectible...

Study smarter with the SolutionInn App


