For each of the following substrates, determine whether an E1 process will require the use of an
Question:
a.
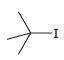
b.
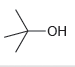
c.
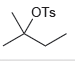
d.
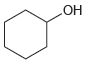
e.
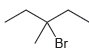
f.
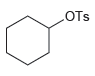
Transcribed Image Text:
-ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a No the leaving group is not OH b Yes ...View the full answer

Answered By

Akshada Ghangale
I have my tutoring experience on other tutoring platform named chegg
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following situations? Determine whether a long or short hedge is appropriate. Justify your answer. a. A firm anticipates issuing stock in three months. b. An investor plans to buy a...
-
For each of the following studies, say whether you would use a t test for dependent means or a t test for independent means. (a) A researcher randomly assigns a group of 25 unemployed workers to...
-
Determine the relaxation time for each of the following medium: (a) Hard rubber ( = 1015 S/m, = 3.1o) (b) Mica ( = 1015 S/m, = 6o) (c) Distilled water ( = 104 S/m, = 80 o)
-
Cadmium is a highly toxic substance with lethal effects on the human body in concentrations greater than 4.4 x 10-mol/L. Cadmium (II) carbonate has a Kp of 5.2 x 10-12 at 25C. Calculate the...
-
Based on, Kerzner, Harold. (2013) Project Management: A Systems Approach to Planning, Scheduling, and Controlling, 11th Ed. John Wiley & Sons. ISBN 978-1118022276.(Chapter 17) and Kerzner, Harold....
-
Ill-conditioning is not only dependent on the matrix of coefficients. This example [Hamming] shows that it can arise from an interaction between the left and right sides of the system. Let be a...
-
Unit Conversion. 1 kilogram (kg) is equal to 2.20462 pounds. Convert \(13 \mathrm{~kg}\) to pounds. Round to three decimal places, if necessary.
-
Spamela Hamderson Inc. reports the following pretax income (loss) for both financial reporting purposes and tax purposes. (Assume the carryback provision is used for a net operating loss.) The tax...
-
work mode : This shows what is correct or incorrect for the work you have completed so far. It does not indicate completion. Return to question Problem 3-40 (LO 3-2, LO 3-3) (Algo) Manny, a...
-
The individual financial statements for Gibson Company and Keller Company for the year ending December 31, 2011, follow. Gibson acquired a 60 percent interest in Keller on January 1, 2010, in...
-
What are fossil fuels?
-
How are coal, oil, and natural gas formed?
-
Consider an economy with just one technique available for the production of each good, food and cloth: a. Supposing land is unlimited but labor equals 100, write and sketch the production possibility...
-
The paper by McCabe and Thiele (1925) is a classic in chemical engineering. Read it. Write a one-page critique of the paper.
-
The APB has made its standards mandatory. Discuss the difficulties associated with publishing a mandatory set of standards for internal auditing.
-
Explain the term shareholder value and describe the role of EVA in measuring and delivering shareholder value.
-
Discuss the importance of non-financial measures of performance in managing a business and the way in which the balanced scorecard attempts to integrate financial and non-financial measures.
-
Define the following: a. Heavy key b. Heavy non-key c. Sandwich component d. Optimum feed stage e. Minimum reflux ratio
-
Suppose that (n) of the points given to the closest-pair algorithm are covertical. Show how to determine the sets P L and P R and how to determine whether each point of Y is in P L or P R so that the...
-
A survey of 70 college freshmen asked whether students planned to take biology, chemistry, or physics during their first year. Use the diagram to answer each question. How many of the surveyed...
-
How strong a base would you expect a Grignard reagent to be? Look at Table 8.1, and then predict whether the following reactions will occur as written.?(The p K a of NH 3 is 35.) (a) CH 3 MgBr + H ?...
-
How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuteratedcompound? Br , CHH-H CHCH2CH
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
Tax Accounting for Mergers and Acquisitions: Tax accounting for mergers and acquisitions (M&A) involves navigating complex regulations and optimizing tax strategies to maximize value for the involved...
-
What is one of the differences between prime costs and conversion costs in manufacturing? A) Only conversion costs include direct labor. B) Only prime costs include manufacturing overhead. Only...
-
MTN H+ X 2024 rt 1 of 1 - 100% 6:46 sakai.ug.edu.gh/porta 73 Question 26 of 40 The basis of the management control function is A. decision making. B. planning. OC. organizing. D. accounting...

Study smarter with the SolutionInn App


