How many signals do you expect in the 13 C NMR spectrum of each of the following
Question:
a.
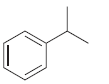
b.
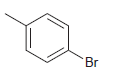
c.
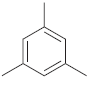
d.
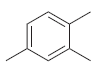
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a 6...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many signals do you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) Geraniol Isolated from roses and used in perfumes . NH2 Dopamine A neurotransmitter that is...
-
Consider the following compound: (a) How many signals do you expect in the 1 H NMR spectrum of this compound? (b) Rank the protons in terms of increasing chemical shift. (c) How many signals do you...
-
Dimethylformamide (DMF) is a common solvent: (a) The 1 H NMR spectrum of DMF exhibits three signals. Upon treatment with excess LAH followed by water, DMF is converted into a new compound that...
-
Solve each system. If a system is inconsistent or has dependent equations, say so. -5x + 2y = -4 6x + 3y = -6
-
Assume the role of a social science researcher in charge of developing a new crime prevention program targeted toward juveniles in the K-12 school system. Review current crime prevention programs...
-
Elite Apparel Inc. is considering two investment projects. The estimated net cash flows from each project are as follows: Each project requires an investment of $900,000. A rate of 15% has been...
-
Think of an idea for a new company and prepare a corporate charter for your business. You can find sample charters online. For extra credit, do one charter for your state and one for Delaware. Using...
-
Doug's Custom Construction Company is considering three new projects, each requiring an equipment investment of $22,000. Each project will last for 3 years and produce the following net annual cash...
-
how to i get the total SF of walls and celing and if room is 10' by 12' with an 8' celing height, it would have two walls that are 10x8 ( Total of 160SF) and two walls that are 12 x 8' ( Total of 192...
-
As someone who came of age in the "post e-mail" world of blogs, wikis, social networks, and other Web 2.0 technologies, you were rather disappointed to find your new employer solidly stuck in the age...
-
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29 a. b. OH HO. HO m/z = 126.0315 m/z = 126.1404
-
Draw a Frost circle for the following cation, and explain the source of instability of this cation.
-
On January 1, 2021, Ameen Company purchased major pieces of manufacturing equipment for a total of $36 million. Ameen uses straight-line depreciation for financial statement reporting and MACRS for...
-
Complete two deeds: one quitclaim deed and one joint tenancy deed. Assume that both of the clients below, Martha and Mary, are individual clients of your law firm (Dewey, Chetam & Howe). The attorney...
-
Interview an older male you know well about what it means to grow up as a male in their culture. a, Who did you interview? b. What were they told about growing up as a male in their culture? c. How...
-
Select and describe any (your choice) recent large sporting or a local/regional commercial event that you are familiar with - maybe it's an event that you were went to growing up. Answer the...
-
A car travels north at 29.3 m/s for 10.7 min. It then travels south at 44.7 m/s for 21.9 min. Instruction: enter your responses below using 3 significant digits using scientific notation (0.00E#) a....
-
A pinball bounced around its machine before coming to rest between two bumpers. Before coming to rest, the ball's displacement was recorded as a series of vectors. All the angles are measured...
-
The Wheeling Tire Company produced a batch of 5000 tires that includes exactly 200 that are defective. a. If 4 tires are randomly selected for installation on a car, what is the probability that they...
-
Define deferred revenue. Why is it a liability?
-
Which of the following alkenes would yield the same alcohol from either oxymercuration-reduction or hydroboration--oxidation, and which would give different alcohols? Explain. (a) cis-2-butene (b)...
-
Give the products (if any) expected from the treatment of each of the following compounds with ozone followed by dimethyl sulfide (a) (b) 2-methylpentane CH2
-
Give the products (if any) expected when the compounds in Problem 5.13 are treated with ozone followed by aqueous hydrogen peroxide.
-
A retainer fee received from a client for future legal representation is an example of a(n) a. accrued revenue Ob. accrued expense Oc. unearned revenue Od. prepaid expense
-
8. (LO 3) You are preparing an analysis of vendors and purchase orders and have transferred data from the ERP system for all purchase orders in the past year and the data for all vendors. You have...
-
Select all that apply Munster Inc. debits Deferred Revenue and credits Service Revenue. As a result of this adjusting entry, Munster's: (Select all that apply.) Multiple select question. liabilities...

Study smarter with the SolutionInn App


