How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
Question:
a.
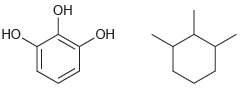
b.
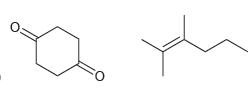
Transcribed Image Text:
Он Но. но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a b OH ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29 a. b. OH HO. HO m/z = 126.0315 m/z = 126.1404
-
How would you distinguish between each pair of compounds using mass spectrometry? a. b. N.
-
How would you distinguish between the following compounds using 13 C NMR spectroscopy?
-
One end of a light, elastic string, of natural length 1.2m and modulus of elasticity 32N, is attached to a fixed point, B. A particle, P, of mass 1.5 kg, is then attached to the other end of the...
-
On July 1,2015, Walter Allen Inc. purchased 6,000 shares of the outstanding common stock of Piaffe Corporation at a cost of $140,000. Piafee had 30,000 shares of outstanding common stock. Assume the...
-
Basis for Zero Trace Matrices Show that Is a basis for the subspace of all 2 x 2 matrices in M22 with zero trace? {lo -91-0 J]- 0)
-
What is the drawback of the coefficient?
-
On August 1, 2010, Sietens Corporation had the following account balances: Raw Material Inventory (both direct and indirect) .... $72,000 Work in Process Inventory ............. 108,000 Finished...
-
What are the advantages and disadvantages of a partnership, and why would you select a sole partnership?
-
Markus Company's common stock sold for $2.75 per share at the end of this year. The company paid a common stock dividend of $0.55 per share this year. It also provided the following data excerpts...
-
The following are mass spectra for the constitutional isomers ethylcyclohexane and 1, 1-dimethylcyclohexane. Based on likely fragmentation patterns, match the compound with its spectrum. 100 100 80 -...
-
An aldehyde with molecular formula C 4 H 6 O exhibits an IR signal at 1715 cm 1 . (a) Propose two possible structures that are consistent with this information. (b) Describe how you could use 13 C...
-
Polly has formed an agreement with a French company to produce and sell her scarves in their 10 stores across Europe. The scarves are Polly's original designs and are of different colors and fabrics....
-
You are a junior auditor working in Big-4 Auditing Limited, a world-wide auditing firm. Recently the CEO of Norden acquire external audit service from Big-4 Auditing Limited. As the CEO of Norden...
-
Stevens IT requires users to choose passwords that consist of exactly 10 capitalized letters, which are stored in hashed form using either SHA-1 or SHA-256. Eve compromised Stevens' authentication...
-
Classification of items as per Cost of quality and determine the total amount on each category under appropriate types: Razor Ltdwants to improve its quality and has the following operating data for...
-
Show a Turing Machine M that decides the following language. E={yyy {0, 1}* } Please 1. Describe in English the idea how it works. 15 points 2. Show all the transitions. 15 points
-
1. What is the meaning of the statement "Leadership is a relationship"? 2. Are leaders born or made? Both? 3. Why is credibility "the foundation of leadership"?
-
Suppose you are part of a trade expedition and there are 15 people in your party (including yourself). Your final destination lies across the desert, and so you must hire out camels to carry all of...
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Open the computer molecular model titled "1-Bromo [2.2.1] bicycloheptane LUMO" at the book's website for the lowest unoccupied molecular orbital (LUMO) of this compound. Where is the lobe of the LUMO...
-
In the previous problem and the associated molecular model at the book's website, you considered the role of HOMOs and LUMOs in an SN2 reaction. (a) What is the LUMO in an SN1 reaction and in what...
-
SN2 reactions that involve breaking a bond to a chirality center can be used to relate configurations of molecules because the stereochemistry of the reaction is known. (a) Illustrate how this is...
-
Assertions about presentation and disclosure typically include: O cutoff, presentation, and disclosure. O cutoff, accuracy, and valuation. completeness, accuracy and valuation, and classification...
-
A client receives ISOs with an exercise price of $24 when the stock is trading at $24. The client decides that she'd like to exercise these options two years after the date of the grant. At that...
-
If Tyrol Willow, the owner of Willow Hardware, reports the following accounts and amounts, the missing amount of Equipment is: Cash $ 16,000 Equipment $ ? Accounts payable 8,000 Wages expense 24,000...

Study smarter with the SolutionInn App


