Identify which of the following monomers would be most reactive toward cationic polymerization. OAc CN CI
Question:
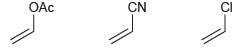
Transcribed Image Text:
OAc CN CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
least r...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify which of the following monomers would be most reactive toward anionic polymerization: CH3 OAc CN
-
Which of the following monomers might you expect would lead to a conducting polymer? a. b. H2C==CHCH3 --,
-
Which of the following monomers might you expect to lead to a conducting polymer? a. CH3CH==CHCH3 b. CH CC CH
-
Bruceton Farms Equipment Company had goodwill valued at $80 million on its balance sheet at year-end. A review of the goodwill by the company's CFO indicated that the goodwill was impaired and was...
-
Use the following information for The Windshield Helper, a retail merchandiser of auto windshields, to compute the cost of goods sold: Web Site Maintenance...
-
Laser World reports net income of $550,000. Depreciation expense is $40,000, accounts receivable increases $10,000, and accounts payable decreases $20,000. Calculate net cash flows from operating...
-
In a survey of U.S. adults, 77% said are willing to share their personal health information to advance medical research. You randomly select 500 U.S. adults. Find the probability that the number who...
-
Sales for Hanebury Corporations just-ended year were $12 million. Sales were $6 million 5 years earlier. a. At what rate did sales grow? b. Suppose someone calculated the sales growth for Hanebury in...
-
On January? 2, 2019, Kaiman Corporation acquired equipment for $ 600000. The estimated life of the equipment is 5 years or 80000hours. The estimated residual value is $ 50000. What is the balance...
-
Chick-fil-A Create a 10- to 12-slide presentation with detailed speaker notes and visuals on every slide that includes the following elements: An analysis of the organizations current culture (e.g.,...
-
Nitroethylene undergoes anionic polymerization so rapidly that it is difficult to isolate nitroethylene without it polymerizing. Explain.
-
Draw a region of an alternating copolymer constructed from vinyl chloride and ethylene.
-
Read about "William Shakespeare's Sonnets" 1. How does Shakespeare deal with the theme of the destructive power of time? Discuss the images he uses. What is his message about time?
-
Assume that none of the fixed overhead can be avoided. However, if the robots are purchased from Tienh Inc., Jobs can use the released productive resources to generate additional income of $375,000....
-
How are prices being set on stock markets? For your explanation, start by (i) stating the optimization principle that reflects the pricing criteria, then (ii) provide your own (!) example of a...
-
Royal Industries had declared a dividend or Rs 5 per share last year . Dividend is expected to grow at 8% p.a. for next three years, and thereafter at 5% in perpetuity. Required return for equity...
-
The implementation of a Central Counterparty (CCP) is an important measure to increase the efficiency of the capital markets. Please (i) explain the basic concept of a CCP and how it increases the...
-
explain the concept of entropy production in irreversible thermodynamic processes, and how it relates to the second law of thermodynamics ?
-
How are uncollectible accounts related to fraud risks?
-
Give the structural formulas of the alkenes that, on ozonolysis, give: a. (CH3)2C=O and CH2=O b. Only (CH3CH2)2C=O c. CH3CH=O and CH3CH2CH=O d. O=CHCH2CH2CH2CH=O
-
Give correct names for the following amines: (a) (b) (c) (d) (e) (f) NH, CH3 CHCHCH3 NHCH3 NH2 ik CH3 NH NH2 NH CHO
-
Show how you would accomplish the following synthetic conversions. (a) Benzyl bromide benzylamine (b) 1-bromo-2-phenylethane 3-phenylpropan-1-amine (c) Pentanoic acid pentan-1-amine (d) Pentanoic...
-
Show how you would prepare the following aromatic amines by aromatic nitration, followed by reduction. You may use benzene and toluene as your aromatic starting materials. (a) Aniline (b)...
-
1. Under 100% sales assumption, using absorption coating, the COGS is calculated by adding Dorect Material (DM), Direct Labor (DL), and Fixed Overhead (FOH) a) True b) False 2. Under 100% sales...
-
. For the rest of questions , refer to the code on the following. Assume both classes are saved in appropriately named files, and are in the same folder. public class Room { int width; int len...
-
Planning quantities Sales $30Q Cost of goods sold $16Q Salaries expense $5,000 per month Utilities expense $250 + $1Q Rent expense $2,100 per month Misc expense $600 + $3Q Actual Results Actual Sales...

Study smarter with the SolutionInn App


