In each compound below, two protons are clearly identified. Determine which of the two protons is more
Question:
(a)
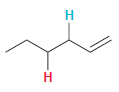
(b)
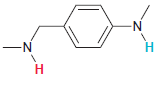
(c)
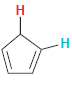
(d)
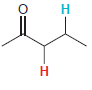
(e)
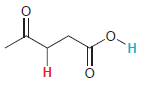
(f)
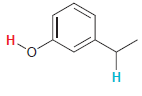
Transcribed Image Text:
Н Н -N Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a ...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
PLS In the Loan Calculator worksheet, the formula in cell D8 should calculate the total cost of the loan. 109 2/3 27. Bao wants t atas Table Get External Data Refresh All Stocks Geogra... L Sort...
-
Which of the following compounds is more easily decarboxylated? CH2CH2 or
-
Which of the following compounds is more likely to exhibit activity as a tranquilizer? CH3 CH CH O or CH C
-
In Exercises, find the limit. x-4 lim x-00x + 1
-
Suppose the risk free rate of return is 6%, maturity risk premium is 2%, inflation premium is 4%, the default risk on similar debt is 3%, and the liquidity premium is 2%. What is the nominal interest...
-
List all the symmetry operations of each of the molecules in Prob. 12.2. In problem 12.2 (a) H2S; (b) NH3; (c) CHF3; (d) HOCl; (e) 1,3,5-trichlorobenzene; (f) CH2F2; (g) CHFClBr.
-
Compute and interpret price and usage variances for material, labour, and overhead inputs.
-
PRIME Inc. purchased 80% of the common shares of SQUARE Inc. for $5,000,000 on January 1, 2017. On that date, SQUARE Inc.'s Common Shares and Retained Earnings were valued at $3,440,000 and...
-
Question 15, 4.3.67-BE Part 1 of 5 HW Score: 68.42%, 13 of 19 points O Points: 0 of 5 he amount (in billions) that the government spents on an ongoing project can be approximated x) 0.186+1.306 In...
-
Snowden Industries produces two electronic decoders, P and Q. Decoder P is more sophisticated and requires more programming and testing than does Decoder Q. Because of these product differences, the...
-
Nitrogen and sulfur are neither in the same row nor in the same column of the periodic table. Nevertheless, you should be able to identify which proton below is more acidic. Explain your choice: .S....
-
Ascorbic acid (vitamin C) does not contain a traditional carboxylic acid group, but it is, nevertheless, still fairly acidic (pKa = 4.2). Identify the acidic proton, and explain your choice using...
-
Pandora Internet Radio by Pandora Media is a streaming music service. Its free advertising-supported radio service was first launched in 2005. Pandora users streamed 20.03 billion hours of Internet...
-
What the title of the THORN symbolize and why William Wordsworth chose this title.
-
What steps are involved when monitoring performance? Give three (3) good key performance indicators in relation to your role in the organisation. 9. What are the different ways an organisation can...
-
he walking trip permission form described in the text is used to cover: J O any trip taken by the children during the year O a specific walking trip taken around the neighborhood O a specific field...
-
Construct a study guide for Introduction to Law examinations for semester one in university
-
a) Explain the various Health Indicators as enshrined in the health sector. (9 marks) b) Assuming an overlapping model and given the following characteristics:- The agents work only in their first...
-
A private not-for-profit entity applies for and receives status as a 501(c)(3) organization. What is the importance of this tax-exempt status?
-
Which internal control principle is especially diffi cult for small organizations to implement? Why?
-
If one were to try to draw the simplest Lewis structure for molecular oxygen, the result might be the following However, it is known from the properties of molecular oxygen and experiments that O2...
-
When ethane is chlorinated, 1,1-dichloroethane and 1,2-dichloroethane, as well as more highly chlorinated ethanes, are formed in the mixture (see Section 10.3A). Write chain reaction mechanisms...
-
(a) What percentages of 1-chloropropane and 2-chloropropane would you expect to obtain from the chlorination of propane if 1( and 2( hydrogen atoms were equally reactive? (b) What percentages of...
-
Brooklyn Inc. offers boat tours down the Hudson River. The company has signed a lease for a tour boat. The company follows ASPE. The terms of the lease are as follows: Lease Term begins on January 1,...
-
A company uses the percent of sales method to determine its bad debts expense. At the end of the current year, the company's unadjusted trial balance reported the following selected amounts: Accounts...
-
At the end of 2020, Payne Industries had a deferred tax asset account with a balance of $45 million attributable to a temporary book-tax difference of $180 million in a liability for estimated...

Study smarter with the SolutionInn App


