In each of the following cases, identify whether the reagent shown is suitable to accomplish the task
Question:
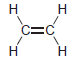
(a) To protonate
using H2O
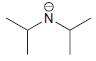
(b) To protonate
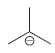
using
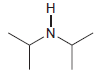
(c) To deprotonate
using
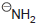
(d) To protonate
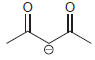
using H2O
(e) To protonate
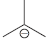
using H2O
(f) To protonate
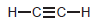
using
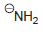
Transcribed Image Text:
OZ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a Yes because a negative charge on an oxygen atom will be more stable than a negative ch...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following cases, calculate the nominal exchange rate assuming purchasing power parity holds. a. A bottle of wine sells for $16 in the United States and 10 in France. b. A book sells...
-
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (a) Cr2 (SO4)3(aq) + (NH4)2CO3(aq) (b) Ba...
-
In each of the following cases draw the structure of an alkyl halide that will undergo an E2 elimination to yield only the indicated alkene. a. b. c. d. ? = E2 E2
-
In Exercises show that the function y = (x) is a solution of the differential equation. y = 4e-x y" - y = 0
-
In the Continuing Payroll Problem A, presented at the end of succeeding chapters, you will gain experience in computing wages and salaries and preparing a payroll register for Kipley Company, Inc., a...
-
(a) Use (15.24) to show that (b) Use the result of (a) to verify that Err
-
Prepare a statement of estimated cash receipts and disbursements for October 2007 for the Aquarius Company, which sells one product, herbal soap, by the case. On October 1, 2007, part of the trial...
-
Items 1 through 16 represent a series of unrelated statements, questions, excerpts, and comments taken from various parts of an auditors working paper file. Below is a list of the likely sources of...
-
Question 35 Linear Functions, Determining The Equation Give termine the Linear Equation for each of the following situ ercept Form. ve the equation of the line passing through the point ,42) that is...
-
The adjusted trial balance for Renfro Company is given in E3.17. Instructions Prepare the income and owners equity statements for the year and the balance sheet at August 31. E3.17 The trial balances...
-
As we will learn in Chapter 21, treating a lactone (a cyclic ester) with sodium hydroxide will initially produce an anion: This anion rapidly undergoes an intramolecular proton transfer, in which the...
-
Identify and name the parent in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i)
-
Mole numbers are conserved in the species continuity equation.
-
Defining Constructivist Theorists and Their Connections Begin by defining and summarizing the key ideas and contributions of the following Constructivist theorists: Jean Piaget Lev Vygotsky Jerome...
-
For each call to the following method, indicate what console output is produced: public void mystery2 (int n) { if (n = 1) { System.out.print(n); } else { mystery2(n 2); System.out.print(", + n); } }...
-
b. What will the predominant species in H2CO3 at pH of 7.5? :0: c. What would be the pH of a buffer that is 1.5 M in H2CO3 and 1.0 M in NaHCO3? [NaHCO3] [H2CO3] pH = pKa + log PH = 6.5 + log 1.0 1.5...
-
Imagine a world where every sound a person utters creates a visible brushstroke in the air, painting a real-time portrait of their emotions. What kind of art movements or social dynamics would emerge...
-
To what extent does the structure of a language, specifically its grammar and vocabulary, influence the way speakers of that language perceive and categorize the world around them, and conversely,...
-
What is an in-kind donation? How are in-kind donations reported?
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
Propose a reasonable mechanism for the following reaction. OH
-
Propose a reasonable mechanism for the following reaction. OH cat. H PO EtOH
-
Vicinal halo alcohols (halohydrins) can be synthesized by treating epoxides with HX. (a) Show how you would use this method to synthesize 2-chlorocyclopentanol from cyclopentene. (b) Would you expect...
-
Dr. Minn is selling her physical therapy practice after owning the practice for 20 years. Dr. Linn is going to buy the radiology practice at a higher price because Dr. Minn has over 500 patients and...
-
Dr. Tier owns a non-profit agency in the local community that provides food, shelter, and counseling for disadvantaged youth and families. At the end of the year, the non-profit agency's difference...
-
Comparative financial statements for Weller Corporation, a merchandisi ompany, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total of...

Study smarter with the SolutionInn App


