In some circumstances, dehydrogenation is observed. Dehydrogenation involves the loss of two hydrogen atoms (the reverse of
Question:
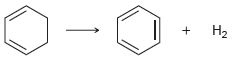
Figure 18.1
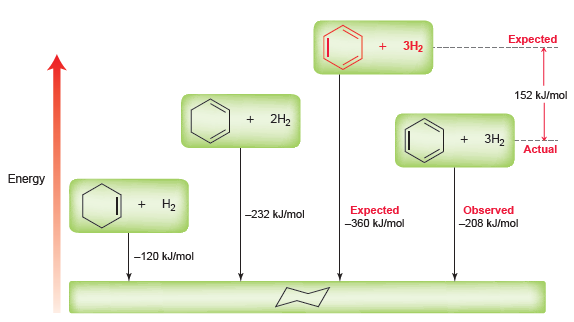
a.
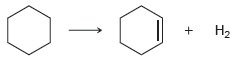
b.
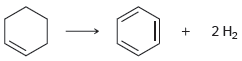
c.
Transcribed Image Text:
Expected ЗНа 152 kJ/mol + 2H2 + ЗН2 Actual Energy | + H2 Expected -360 kJ/mol Observed -232 kJ/mol -208 kJ/mol -120 kJ/mol H2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a H has a posi...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The dehydrogenation of butane to trans-but-2-ene has (H( = +116 kJmol (+27.6 kcal/mol) and (S( = +117 J/kelvin-mol (+28.0 cal/kelvin-mol). (a) Compute the value of (G( for dehydrogenation at room...
-
At high temperatures, alkanes can undergo dehydrogenation to produce alkenes. For example: This reaction is used industrially to prepare ethylene while simultaneously serving as a source of hydrogen...
-
The Rice-Herzfeld mechanism for the dehydrogenation of ethane is specified in Section 23.1, and it was noted there that it led to first-order kinetics. Confirm this remark, and find the...
-
The specifications for the water supply system of the Sears Tower in Chicago require that 100 gpm of water be pumped to a reservoir at the top of the tower, which is 340 m above street level. The...
-
For the data given below, classify the soils according to the USCS. For each soil, give both the letter symbol and the narrative description. (a) 65% material retained on No. 4 sieve, 32% retained on...
-
Consider the function f (x) = 3(x + 1)2 - 4. a. Find f (5). b. Find f (n). c. Find f (x + 2). d. Use your calculator to graph y = f (x) and y = f (x + 2) on the same axes. How do the graphs compare?
-
Oswego Corporation reported net income $24,000; net sales $400.000; and average assets \($600,000\) for 2002. What is the 2002 profit margin? (a) 6%. (b) 12%. (c) 40%. (d) 200%.
-
Lowery, Inc. purchased new plant equipment on January 1, 2011. The company paid $920,000 for the equipment, $62,000 for transportation of the equipment, and $10,000 for insurance on the equipment...
-
Required Information [The following information applies to the questions displayed below.] A company makes the payment of a one-year insurance premium of $4,464 on March 1, 2019. -1. Use the...
-
Auditors for the Internal Revenue Service (IRS) scrutinize income tax returns after they have been prescreened with the help of computer tests for normal ranges of deductions claimed by taxpayers....
-
Compound A has molecular formula C 8 H 8 . When treated with excess Br 2 , compound A is converted into compound B, with molecular formula C 8 H 8 Br 2 . Identify the structures of compounds A and B....
-
Predict whether each of the following compounds should be aromatic. a. b. c.
-
Office Plus is a retail business that sells office equipment, furniture, and supplies. Its credit purchases and purchases returns and allowances for September are shown on the next page. The general...
-
Welcome to week 5. This week we will be discussing an important topic, teacher evaluations. There has been a big push to count the teacher evaluation as a determining factor toward incentive pay...
-
Anna is on a wagon and Amy is pulling down and to the right with a force of 1 0 0 N at an angle of 2 5 degrees below the horizontal. Anna and the wagon have a mass of 5 5 kg and they are moving at a...
-
A company reports the following for one of its products. Direct materials standard (5 pounds @ $2 per pound) Actual direct materials used (AQ) Actual units produced Actual cost of direct materials...
-
11. During the course of his audit of the financial statements of a corporation for the purpose of expressing an opinion on the statements, an auditor is denied permission to inspect the minutes of...
-
What is a composite material? Give two examples of a natural composite material and two examples of a manufactured composite.
-
You have data for many years on the average price of a barrel of oil and the average retail price of a gallon of unleaded regular gasoline. If you want to see how well the price of oil predicts the...
-
Starr Co. had sales revenue of $540,000 in 2014. Other items recorded during the year were: Cost of goods sold ..................................................... $330,000 Salaries and wages...
-
Draw tetrahedral representations of the two enantiomers of the amino acid cysteine, HSCH 2 CH (NH 2 ) CO 2 H, and identify each as R or S.
-
The naturally occurring form of the amino acid cysteine (Problem 9.48) has the S configuration at its chirality center. On treatment with a mild oxidizing agent, two cysteines join to give cystine, a...
-
Which of the following pairs of structures represent the same enantiomer, and which represent different enantiomers? (b) Br Br C (a) CN - N - - r H3CC-CN Br CN (c) (d) CH C g H- H2N CH - -H -...
-
A: December 31, 2021, Southern Corporation reported the stockholders' equity accounts shown here (with dollar amounts in milions, except per-share amounts) (Click the icon to view the data)...
-
Part B Short Answer Questions Generally, each Part B short answer question would take about 250 words to answer adequately. Topic 4 (Foreign currency transactions) Sydney Ltd is an Australian company...
-
Concerning a RAL, which of the following is a true statement that needs to be discussed with the tax client? Question 16Select one: a. That the RAL is an "advanced" income tax refund b. A disclosure...

Study smarter with the SolutionInn App


