Kodel is a synthetic polyester with the following structure. (a) Identify what monomers you would use to
Question:
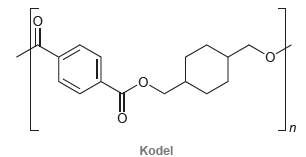
(a) Identify what monomers you would use to make Kodel.
(b) Would you use acidic conditions or basic conditions for this polymerization process?
Transcribed Image Text:
in Kodel
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a b Ac...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Kodel is a polyester with the following structure: From what two monomers is it made? -O-CH CH2-O
-
The compound benzenc has only one type of carbon-carbon bond, and this bond has a length intermediate between that of a single bond and a double bond. Draw a resonance structure of benzene that,...
-
Amino acids are biological compounds with the following structure, where the R group can vary. The structure and biological function of amino acids will be discussed in Chapter 25. Identify the total...
-
Use the present value tables in Appendix A and Appendix B to compute the NPV of each of the following cash inflows. a. $89,000 received at the end of six years. The discount rate is 4 percent. b....
-
True Fit Shoe Company makes loafers. During the most recent year, True Fit incurred total manufacturing costs of $21,900,000. Of this amount, $2,600,000 was direct materials used and $14,800,000 was...
-
Mega Screens, Inc., reports sales revenue of $2,700,000, cost of goods sold of $1,500,000, and income tax expense of $140,000 for the year ended December 31, 2012. Selected balance sheet accounts are...
-
In a survey of U.S. adults, 43% said they would favor allowing people to carry guns in more places. You randomly select 100 U.S. adults. Find the probability that the number who favor allowing people...
-
Optima Company is a high-technology organization that produces a mass-storage system. The design of Optima's system is unique and represents a breakthrough in the industry. The units Optima produces...
-
please show all work. FIN 301 Ex Problem #1 (Ratios): Tom's Fresh Market is a family owned produce and garden market servicing the northwest side of Chicago. Founded in 1980, Tom's provides the...
-
Hospital personnel routinely examine patient records for error, such as incomplete insurance information, on incomplete patient history, or missing/incomplete medical records. On average, about 250...
-
Explain why vinyl shower curtains develop cracks over time.
-
Polyformaldehyde, sold under the trade name Delrin, is a strong polymer used in the manufacture of many guitar picks. It is prepared via the acid-catalyzed polymerization of formaldehyde. (a) Draw...
-
Determine the breakeven point for each plant. Hambry Enterprises produces a component for recycling uranium used as a nuclear fuel in power plant generators in France and the United States. Use the...
-
Why are commercial banks owned by shareholders a disadvantage to customers?
-
Your firm represents Jimmy Jones. He and his best friend, Bobby Smith, are both 20-year-old high school dropouts. They have held several part-time jobs in the past but are currently unemployed. Last...
-
Find the anti-derivative class (do not forget the +C) for Evaluate the definite integral L 1 x dx= 1+x6 x dx= 1+x6
-
a) Explain how financial ratio analysis of a firm's projected cash flow budget could be efficiently used by its managers for financial planning. (b) Explain why creating budgets and other financial...
-
DIY problem: Charge Q is distributed uniformly inside a sphere of radius R. Calculate and sketch electric field E, a) outside the sphere & b) inside the sphere. (6) (a) Surface (a) encloses all of...
-
Jeff Jacobs is a junior accountant with the public accounting firm of Maxwell and King LLP. Jeff was assigned to do the audit of Astor Electronics Inc. The in-charge auditor informed Jeff that, based...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
Several additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitro compounds and the Gabriel synthesis leave the carbon chain unchanged. Formation...
-
(a) Guanidine (shown) is about as strong a base as hydroxide ion. Explain why guanidine is a much stronger base than most other amines. (b) Show why p-nitroaniline is a much weaker base (3 pKb units...
-
Show how you would synthesize the following compounds starting with benzene, toluene, and alcohols containing no more than four carbon atoms as your organic starting materials. Assume that para is...
-
JavaFx. Drop a yellow disc at column (0 ?? 6) : 6 0 l 2 3 4 5 6 01(0) (.))))) 0)) ))) 0),) D) 0)), )3 The yellow player won. Congratulations. Create a program that allows the user to create array...
-
IMC Pty Ltd produces plastic ice cube trays in two processes: heating and stamping. All materials are added at the beginning of the Heating Department process, and IMC Pty Ltd uses the...
-
1. EX.22.05.ALGO eBook Show Me How Question Content Area Production budget Healthy Habits Inc. produces a Bath version and a Gym version of its popular electronic scale. The anticipated unit sal...

Study smarter with the SolutionInn App


