Melphalan is a chemotherapy drug used in the treatment of multiple myeloma and ovarian cancer. Melphalan is
Question:
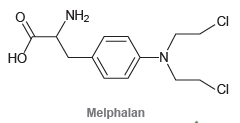
Transcribed Image Text:
NH2 .CI но -N CI Melphalan
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
HO HO NH melph...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Pharmacokinetics is the study of the rates of absorption and elimination of drugs by organisms. In most cases, elimination is slower than absorption and is a more important determinant of...
-
The sodium salt of valproic acid is a drug that has been used in the treatment of epilepsy. (Valproic acid is a name used in medicine.) (a) Give the substitutive name of valproic acid. (b) Give the...
-
Ambien TM is a sedative used in the treatment of insomnia. It was discovered in 1982 and brought to market in 1992 (it takes a long time for new drugs to undergo the extensive testing required to...
-
In Exercises 1318, find the average rate of change of the function from x 1 to x 2 . f(x)=x from x = 4 to x = 9
-
Daily in any healthcare organization there are many challenges that employees and management staff must confront. Some of these challenges may occur due to governmental regulation which may not be...
-
Let Compute the inverse of C, C-1, by forming the 4 Ã 8 matrix [C | I4] and row-reducing (Theorem CINM). Then use a calculator to compute C-1 directly. 1 131 2 -4-1 2 2 0-4 5 c=11 410
-
What are we forgetting when we say that by increasing return on equity, the leverage effect of debt cannot increase value?
-
Prepare the monthly income statement, monthly statement of owners equity, and the balance sheet at August 31, 2014, for Krishna Cleaning Company from the data provided in the adjusted trial balance...
-
Question 2 (Marks: 30) The following information of Mimosa General Dealers for July 2021 has been provided to you: . The debit balance per the bank account at 31 July 2021 was R6 250. . Bank charges...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
What reagents would you use to accomplish a substitution with retention of configuration; for example? OH SH (R)-2-Butanol (R)-2-Butanethiol
-
List the systematic name and common name for each of the following compounds: (a) (b) (c) (d) (e) CI Br
-
Find the position of the center of mass of the system of the sun and Jupiter. (Since Jupiter is more massive than the rest of the planets combined, this is essentially the position of the center of...
-
dentify a situation you have been in a team. What sort of team was it and what was your task as a team? Explore group action within your team. Was social facilitation or social loafing more evident?...
-
For the following systems, evaluate the boundary condition at the interface as mol% and wt%. a.) Air-water system at 75F with P = 760 mmHg and psat for water is 22.5 mmHg. Determine the composition...
-
Jamison Company has two service departments and two producing departments. Square footage of space occupied by each department follows: Custodial services General administration Producing Department...
-
The following transactions pertain to an ALBANY COMPANY branch's second month's operations in June, 2020: 1. The HOME OFFICE sent P14,400 cash to the BRANCH. 2. The HOME OFFICE shipped inventory...
-
= (a) (2 marks) Let A = {x Zx 3} and B following sets (no explanation required)? i. AUB ii. An B iii. (ANB) UA iv. A\B v. B\A vi. BUBUB {-4, -2,2,4}. What is the cardinality of the (b) (5 marks) Let...
-
Fred says that he ran the Huffman coding algorithm for the four characters, A, C, G, and T, and it gave him the code words, 0, 10, 111, 110, respectively. Give examples of four frequencies for these...
-
Write a paper by answer the following question: Should Recycling Be Mandatory?
-
Vaccenic acid, a constitutional isomer of oleic acid, has been synthesized through the following reaction sequence: Propose a structure for vaccenic acid and for the intermediates A-E. liq. NaUN KUH,...
-
(a) Show the isoprene units in each of the following terpenes. (b) Classify each as a monoterpene, sesquiterpene, diterpene, and so on. CH Zingiberene (from oil of ginger (from oil of celery)...
-
w-Fluorooleic acid can be isolated from a shrub, Dechapetalum toxicarium, that grows in Africa. The compound is highly toxic to warm-blooded animals; it has found use as an arrow poison in tribal...
-
Brooklyn Inc. offers boat tours down the Hudson River. The company has signed a lease for a tour boat. The company follows ASPE. The terms of the lease are as follows: Lease Term begins on January 1,...
-
A company uses the percent of sales method to determine its bad debts expense. At the end of the current year, the company's unadjusted trial balance reported the following selected amounts: Accounts...
-
At the end of 2020, Payne Industries had a deferred tax asset account with a balance of $45 million attributable to a temporary book-tax difference of $180 million in a liability for estimated...

Study smarter with the SolutionInn App


