Methylmagnesium bromide reacts rapidly with ethylene oxide, it reacts slowly with oxetane, and it does not react
Question:
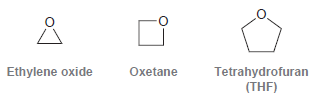
Explain this difference in reactivity.
Transcribed Image Text:
Oxetane Ethylene oxide Tetrahydrofuran (THF)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Ethylene oxide has a high degree of ring strain and readily functions as an electrophile in an S ...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write an equation for the reaction, if any, of p-bromobenzaldehyde with each of the following reagents, and name the organic product. a. Methylmagnesium bromide, then H3O+ b. Methylamine (CH3NH2) c....
-
A compound reacts with methylmagnesium bromide followed by acidification to form the product with the following 1H NMR spectrum. Identify the compound. 6 (ppm) 2 frequency
-
Ethylene oxide, C2H4O, decomposes when heated to give methane and carbon monoxide. C2H4O(g) CH4(g) + CO(g) The following kinetic data were observed for the reaction at 688 K: Find the rate law and...
-
DBU Systems manufactures testing equipment for the communications industry. In developing a new device for maritime communication, the design group has estimated the following unit costs. Metal...
-
Parent Co. purchases 100% of Son Company on January 1, 20X1 when Parent's retained earnings balance is $520,000 and Son's is $150,000. During 20X1, Son reports $15,000 of net income and declares...
-
Steam enters a converging nozzle at 800 kPa and 280 C with negligible velocity. If expansion is isentropic. What is the minimum pressure that can be reached in such a nozzle and what is the...
-
Drop shippers take title to products but do not accept physical possession of them, whereas commission merchants take physical possession of products but do not accept title. Defend the logic of...
-
Let's see whether quadratic voting can avoid the paradox of voting that arose in Table 5.3 when using 1p1v in a series of paired-choice majority votes. To reexamine this situation using quadratic...
-
Decision on Accepting Additional Business Homestead Jeans Co. has an annual plant capacity of 65,900 units, and current production is 45,800 units. Monthly fixed costs are $38,300, and variable costs...
-
In 20X1, its first year of operations, Regal Department Store sells $250,000 of gift certificates redeemable for store merchandise that expire one year after their issuance. With a high degree of...
-
The Williamson ether synthesis cannot be used to prepare tert-butyl phenyl ether. a. Explain why this method cannot be used in this case. b. Suggest an alternative method for preparing tert-butyl...
-
Identify the reagents necessary to accomplish each of the following transformations. >
-
What coarse-grained (plutonic) rock consists of abundant calcic plagioclase plus olivine and pyroxene?
-
1. Create a MySQL database to store the registration data about your customers as well as a login, password, and two security questions for each customer. 2. Implement your design in MySQL, using any...
-
Required: Compute the gross profit percentage in the current and previous years. Are the current - year results better, or worse, than those for the previous year? Compute the net profit margin for...
-
*ANSWER THE REQUIREMENTS IN EXCEL WITH =FORMULATEXT VISIBLE FOR EVERY NUMBER CALCULATED* The following data relate to the operations of Shilow Company, a wholesale distributor of consumer goods:...
-
What does the following function call do to this object? What does the function return? the_list.make_set("pony") the_list front back
-
The following additional information is available about flight 482: Members of the flight crew are paid fixed annual salaries, whereas the flight assistants are paid based on the number of round...
-
A student wants to convert a set of six z-scores (4.2, 1.6, +0.1, +0.6, +1.4, +2.1) to a scale that has a mean of 80 and a standard deviation of 20. a. What is the new score for each of these six...
-
Consider the circuit of Fig. 7.97. Find v0 (t) if i(0) = 2 A and v(t) = 0. 1 3 ett)
-
In step 6 of fatty-acid biosynthesis (Figure), acetoacetyl ACP is reduced stereo specifically by NADPH to yield an alcohol. Does hydride ion add to the Si face or the Re face of acetoacetylACP?...
-
In step 7 of fatty-acid biosynthesis (Figure), dehydration of a ?-hydroxy thioester occurs to give trans-crotonyl ACP. Is the dehydration a syn elimination or an anti elimination? CH3SCOA Acetyl CoA...
-
In step of fatty-acid biosynthesis (Figure), reduction of Trans-crotonyl ACP gives butyryl ACP. A hydride from NADPH adds to C3 of the crotonyl group from the Re face, and Protonation on C2 occurs on...
-
P11.34 Overhead variances; journal entries; closing variance accounts: manufacturer LO11.5 Broome Instruments Company manufactures a control valve used in air-conditioning systems. The firm uses a...
-
What are the problems with the ideal CNN model? A. Requirement of a large and labeled image dataset B. Higher computational cost for training deep CNNs C. Lower computational cost for training deep...
-
Pregunta 10 1 pts El ingenio Mujica labora en INHGEOMIN; el contador de la institucin le informa que una mina en explotacin tiene una produccin anual de $600,000 y se calcula que se agotar en cinco...

Study smarter with the SolutionInn App


