Predict the major product obtained when each of the following compounds is treated with Birch conditions. a.
Question:
a.
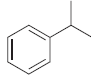
b.
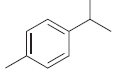
c.
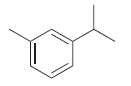
d.
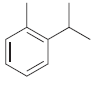
e.
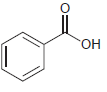
f.
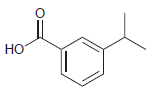
Transcribed Image Text:
OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product obtained when each of the following compounds undergoes a Claisen condensation. (a) (b) (c) OEt OM
-
Predict the major product obtained when each of the following aldehydes is treated with aqueous sodium hydroxide: (a) (b) (c) (d) H. H.
-
Predict the major product obtained when each of the following amines is treated with a mixture of NaNO 2 and HCl: (a) (b) (c) (d) NH2 Z
-
You find a certain stock that had returns of 18 percent, 23 percent, 16 percent, and 9 percent for four of the last five years. If the average return of the stock over this period was 10.3 percent,...
-
On July 11, 1984, Jose Padilla was working as a vacation-relief route salesperson for Frito-Lay. He testified that he made a route stop at Sal's Beverage Shop, where he was told by Mrs. Ramos that...
-
A Vector Space Equation Suppose that, for c E R and in vector space v in vector space V, cv = 0. Then show that either c = 0 or v = 0?
-
Will liquidity premiums tend to rise or fall during a crash? Why?
-
Merry Olde Games produces croquet sets. The company makes fixed monthly payments to the local utility based on the previous years electrical usage. Any difference between actual and expected usage is...
-
Additional information was available after the Financial Statements in Part A were completed. Therefore, critically analyze the effect of each of the following items separately on the performance...
-
The first column shows the number of donuts consumed, while the second and third columns (respectively) show Shanes and Miriams marginal personal use values. Assume that Shanes initial endowment is...
-
Propose a plausible synthesis for the following transformation.
-
Consider the structure of anisole (also called methoxybenzene). In the next chapter, we will discuss whether a methoxy group is electron donating or electron withdrawing. We will see that there is a...
-
What is a significance level? How does a researcher choose a significance level?
-
What are ways a firm can strengthen it's Sustainable Competitive Advantage ( SCA ) position?
-
What are the fives competitive forces that shape strategy? Explain
-
Explain porters competitive strategies focusing on low cost strategy and differentiation strategy with example.
-
Identify a relevant scenario from workplace or a famous case where you can apply and benefit from at least one stakeholder influencing tool as described of the PM Toolbox textbook. The tool(s) that...
-
Describe the competitive positions in the drone industry and assess the opportunities for at least three positions in the industry.
-
The maximum independent set (MIS) of a graph G = (V,E) is the largest set of vertices S V such that for any two vertices u, v S, (u, v) / E; that is, no pair of vertices in S are neighbors. We want...
-
Refer to the Conservation Ecology (Dec. 2003) study of the causes of forest fragmentation, presented in Exercise 2.166 (p. 97). Recall that the researchers used advanced high-resolution satellite...
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) (e) Nal (1 mol) H -acetone GH,H + NaBr Nal (1 mol), CH,NaCl CI CI BrBr (1 mol C4HS2 2 NaBr OH NaH (-H2) Cl Et O...
-
When tert-butyl bromide undergoes SN1 hydrolysis, adding a "common ion" (e.g., NaBr) to the aqueous solution has no effect on the rate. On the other hand, when (C6H5)2CHBr undergoes SN1 hydrolysis,...
-
When the alkyl bromides (listed here) were subjected to hydrolysis in a mixture of ethanol and water (80% EtOH/20% H2O) at 55oC, the rates of the reaction showed the following order: (CH3)3CBr >...
-
American Supply sold merchandise on account to Decker Plumbing on March 31. The sales price was $2,300, and the cost of goods sold was $1,500. Sales were reported immediately, but the cost of goods...
-
As of December 31, 2022, what percentage of JPMorgan Chase Bank, N.A.'s deposits held in domestic offices were insured? Total Deposits: 76.23% of total assets Deposits held in Domestic Offices:...
-
Part 3: Is covered in Module 23. I also have support material in the Module 23 Resources in the applicable weekly area. The roofing company manufactures shingles. Standard Cost Sheet per shingle...

Study smarter with the SolutionInn App


