Predict the position of equilibrium for each of the following reactions: (a) (b) (c) HO NH NH2
Question:
(a)
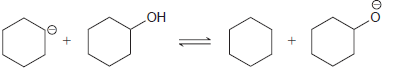
(b)
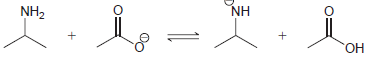
(c)
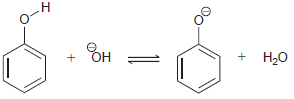
Transcribed Image Text:
HO NH NH2 ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a The rig...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the position of equilibrium for each acid-base reaction below: (a) (b) (c) (d) `H. O + H20
-
Draw the products of each of the following acid-base reactions, and then predict the position of equilibrium in each case: (a) (b) NaH
-
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of...
-
Each student will interview a manager or an employee (who might be a family member, a friend, or an acquaintance) to determine the extent to which the issues raised in the case are represented in his...
-
In the short-run (before P adjusts) version of the IS-LM model, a temporary decrease in government purchases causes the real interest rate to (blank) and output to (blank): a. rise; rise b. rise;...
-
Which of the following functions are homogeneous? Give the degree of homogeneity. (a) x + 3yz; (b) 179; (c) x2/yz3; (d) (ax3 + bxy2)1/2
-
Daphne Kite Company wants a master budget for the next three months, beginning January 1, 2008. It desires an ending minimum cash balance of $5,000 each month. Sales are forecasted at an average...
-
Identify each of the following accounts of Advanced Services Co. as asset, liability, owners equity, revenue, or expense, and state in each case whether the normal balance is a debit or a credit. a....
-
22. Here is a histogram of T-cell velocities in vitro (in microns per minute): Count 350 300 250 200 150 100 50 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Velocity (microns/min) The data represent a...
-
Suppose an individual makes an initial investment of $2500 in an account that earns 7.8%, compounded monthly, and makes additional contributions of $100 at the end of each month for a period of 12...
-
Amphotericin B is a powerful antifungal agent used for intravenous treatment of severe fungal infections. Identify the most acidic proton in this compound: , Amphotericin B NH2
-
As we will learn in Chapter 21, treating a lactone (a cyclic ester) with sodium hydroxide will initially produce an anion: This anion rapidly undergoes an intramolecular proton transfer, in which the...
-
Fenwicke Company began operating a subsidiary in a foreign country on January 1, 2011, by acquiring all of its common stock for LCU 40,000, which was equal to fair value. This subsidiary immediately...
-
What is the first step a company should take when developing a job design and a method analysis and a methods of analysis. Talk with employees for data gathering compare a new job to an existing job....
-
2. (a) (3 points) How many bits are there in a half byte? (b) (3 points) How many distinct patterns can you make with half a byte? 3. (8 points) Write a Python function that takes three numbers as...
-
In a kidnapping scenario, the kidnapper (Guy) would be inclined to kill the kidnappee (Orlando) if Orlando sees Guy's face, because if Orlando were released, he would be able to help the police...
-
You live in a large community, are in recovery from a sexual assault, and attend a PTSD support group near your home. At one of the meetings a former client introduces herself as a new member of the...
-
A not-for-profit entity reports contributed services valued at $38,000. How was the decision made to report these services?
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
Predict the products from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) Hl (excess) Hi H,so,,H20 O MeONa O MeOH, cat. H2SO (1) EtSNa (2) H2o HCl (1 equiv.) MeONa (1)EtON...
-
Provide the reagents necessary to accomplish the following syntheses. (a) (b) (c) (d) MeO MeO SEt SEt
-
Provide reagents that would accomplish the follwing syntheses. (a) (b) HO Glycerol Epichlorohydrin
-
Why do parent companies disregard the legal boundaries of entities, and report consolidated financial statements as a single economic entity? Select answer from the options below to provide useful...
-
Identify a true statement about merit increases in a small business. Multiple choice question. It is an arrangement whereby an employee receives a prescribed share of the business's profits. It is...
-
Price Company assigns overhead based on machine hours. The Milling Department logs 1,800machine hours and Cutting Department shows 3,000 machine hours for the period. If theoverhead rate is $5 per...

Study smarter with the SolutionInn App


