Predict the products for each of the following reactions, and propose a mechanism that explains the formation
Question:
(a)
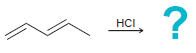
(b)
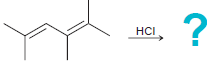
(c)
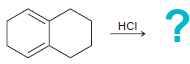
(d)
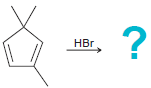
(e)
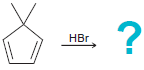
(f)
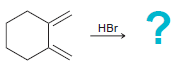
Transcribed Image Text:
HCI HCI HCI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b c d e f C...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products for each of the following reactions: (a) (b) H2 Lindlar's catalyst :? H2 Pt -? Na NH, (1) ? Ni,B Pd Na NH3 (/)
-
Predict the products for each of the following reactions, and in each case, determine which product will predominate: (a) (b) (c) HBr 40C HCI 0C
-
Predict the products for each of the following reactions. In some cases, the reaction produces a new chirality center, while in other cases, no new chirality center is formed. Consider that when...
-
Suppose we want both the sensitivity and specificity to be at least 70%. Use the ROC curve to identify the possible value(s) to use as the cutoff for identifying people with dementia, based on these...
-
What are two advantages of Office 365 compared to the traditional Office?
-
It is an exciting time to be a plant breeder because so many options are available for the development of new types of agriculturally useful plants. Let's suppose you wish to develop a seedless...
-
When the gross profit statements option is chosen, a separate report is generated for each department. (True/False)
-
Identify the factors that are relevant in determining the annual depreciation charge, and explain whether these factors are determined objectively or whether they are based on judgment.
-
What are possible reasons for a pension plan to be underfunded? Select one: a. All of the listed answers b. Actual returns on pension plan assets are less than expected. c. Changes in actuarial...
-
Kinetic behavior of a TRIGA reactor can be modeled using the Point Reactor Kinetics Equations (PRKE?s). The simplified one-group PRKE?s are With initial conditions; reactor temperature T0 = 300K,...
-
Table 2.1 contains information on three techniques for producing $15 worth of bar soap. Assume $15 worth of bar soap means the selling price of soap is $3 per bar and all three techniques produce 5...
-
Suppose Natasha currently makes $50,000 per year working as a manager at a cable TV company. She then develops two possible entrepreneurial business opportunities. In one, she will quit her job to...
-
What are the five components of a full set of financial statements?
-
A manager at Z Ltd. wants to replace an old machine with a new, more efficient machine. New machine: List price 90,000 $ Annual variable expenses 80,000 Expected life in years 5 Old machine: Original...
-
Use the formula for the present value of an ordinary annuity or the amortization formula to solve the following problem. PV $12,000; PMT = $600; n=35; i=? (Type an integer or decimal rounded to three...
-
Q.1. Schneider manufacturing company has following information for March 2019. Budgeted FOH for 10,000 hours $ 30,000 Budgeted FOH for 16,000 hours $ 42,000 FOH applied rate for $ 3 per hour Actual...
-
The function f(x)=x2-x-2 is multiplied by a constant value to create a new function g(x)=af(x). a) If the new function g(x) passes through the point (3, 14), what is the value ofa? b) what are the...
-
Factor. 2x + 12x-54
-
Machines that fill bags with powdered sugar are supposed to dispense 32 ounces of powdered sugar into each bag. Let X = the weight (in ounces) of the powdered sugar dispensed into a randomly selected...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
What products would you expect from the following reactions? CH2 (a) + CHCI3 CH (b) Zn(Cu) CH2I2 H2HH
-
What product would you obtain from catalytic hydrogenation of the following alkenes? CH2H (a) (b) CH "CH
-
What product would you expect from reaction of cis-2-butene with meta-chloro-peroxybenzoic acid? Show the stereo chemistry.
-
Dr. Minn is selling her physical therapy practice after owning the practice for 20 years. Dr. Linn is going to buy the radiology practice at a higher price because Dr. Minn has over 500 patients and...
-
Dr. Tier owns a non-profit agency in the local community that provides food, shelter, and counseling for disadvantaged youth and families. At the end of the year, the non-profit agency's difference...
-
Comparative financial statements for Weller Corporation, a merchandisi ompany, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total of...

Study smarter with the SolutionInn App


