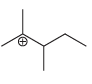
Predict whether each of the following carbocations will rearrange. If so, draw the expected rearrangement using curved
Question:
a.
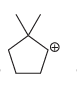
b.
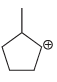
c.
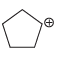
d.
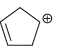
e.
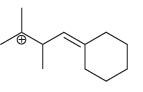
f.
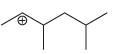
g.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a b c This carbocation is seco...View the full answer

Answered By

Michael Mulupi
I am honest,hardworking, and determined writer
4.70+
72+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether each of the following molecules is polar or non-polar: (a) IF (b) CS2 (c) SO3 (d) PCl3 (e) SF6 (f) IF5
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict whether each of the following compounds is molecular or ionic: (a) B2H6 (b) CH3OH (c) LiNO3 (d) Sc2O3 (e) CsBr (f) NOCl (g) NF3 (h) Ag2SO4.
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
Read the case "The Boys Versus Corporate" and write a short paper related to cultures
-
An importer of television sets from the Far East distributes them throughout the European Union from four warehouses. Shipments are received monthly, and this month's shipment is 120,000 sets. Due to...
-
Information for four separate finance/sales-type lease scenarios is provided as follows: Required Answer the following questions for each separate scenario assuming that the lessee is aware of the...
-
(2-Year Worksheet) On January 1, 2010, Cunningham Company has the following defined benefit pension plan balances.? Projected benefit obligation ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ?$4,500,000 Fair...
-
You are evaluating a project for The Farstroke golf club, guaranteed to correct that nasty slice. You estimate the sales price of The Farstroke to be $440 per unit and sales volume to be 1,200 units...
-
A publicly listed traded company is in financial distress. It is projected to stop paying dividends and is likely to stop trading as a going concern in the near future. Which of the valuation methods...
-
Draw curved arrows for each step of the following mechanism: : : : 0: . : : -O: : - : : : : H20 -
-
Consider the following reaction: The following rate equation has been experimentally established for this process: Rate = k [HO - ] [CH 3 CH 2 Br] The energy diagram for this process is shown below:...
-
Using only the periodic table, determine which bond in each pair is more ionic: (a) HF or HCl (b) OF or CF
-
Anna Drew sells custom cabinets and doors. She receives a 15% commission on the doors she sells and a 7% commission for each cabinet. What would her total commission be if she sold $5,200-worth of...
-
Calculate each account balance. (Enter the balance, along with a "Bal" posting reference on the appropriate side of each T-account.) Supplies 132 Note payable 221 3/8 1,100 3/27 1,000 3/20 1,300 3/5...
-
A local pizza store knows the mean amount of time it takes them to deliver an order is 45 minutes after the order is placed. The manager has a new system for processing delivery orders, and they want...
-
coni nomin Record the following transactions in the General Fund general ledger accounts. (Analyze the effects of the transactions on all other funds and nonfund accounts.) 1. Martinsville ordered...
-
In this assessment, you will work in a group to critically analyse beliefs about children and early childhood practice and creatively reimagine how this might look in practice. This will be in the...
-
When Mendel conducted his famous hybridization experiments, he used peas with green pods and yellow pods. One experiment involved crossing peas in such a way that 25% (or 145) of the 580 offspring...
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
Compound B, an isomer of A (Problem 20.47), is also soluble in dilute HCl. The IR spectrum of B shows no bands in the 3300-3500-cm-1 region. The broadband proton-decoupled 13C spectrum of B is given...
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
Show how you might prepare each of the following amines through reductive amination: (a) (b) (c) NH2 CH3 CH3
-
Aanya is giving away sixteen identical candles as favours at her latest party! Eight guests in total are attending the party. In how many ways can the favours be given out if Aanya wants to ensure...
-
Deliverable #1 (2 Marks) due week 10 for professor review and feedback Select two entity classes from deliverable #3 "Part B" and develop two state diagrams. The classes should have a dynamic nature...
-
The differential equation a3y"" + y" + y' + oy = 0 has at least one solution of the form eax, a ER. True False Y2 If | | = 0, then y and y2 are fundamental set of solutions of the y'2 # differential...

Study smarter with the SolutionInn App


