Predict which of the following compounds is more acidic, and explain your choice. N- -N- -NH2 NH2
Question:
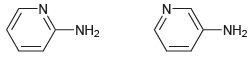
Transcribed Image Text:
N- -N- -NH2 NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Compare the conjugate bases Both are resonance stabilized ...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict which of the following compounds is more acidic. After making your prediction, use the pK a values from the following table to determine whether your prediction was correct. ACID pka...
-
Identify which of the following compounds is more acidic and explain your choice.
-
Identify which of the following compounds is more acidic. Explain your choice. C=C H--
-
Lewis and Associates has been in the termite inspection and treatment business for five years. The following is a list of accounts for Lewis on June 30, 2017. It reflects the recurring transactions...
-
On a foreign exchange market diagram with the dollar price of the British pound on the vertical axis, explain why you draw an upward-sloping supply curve.
-
Calculate the pH of a 0.20 M ammonium acetate (CH3COONH4) solution.
-
The purpose of negative reinforcement as an operant conditioning technique is to ____________. (a) punish bad behavior (b) discourage bad behavior (c) encourage desirable behavior (d) offset the...
-
Wabash Corp. just completed another successful year, as indicated by the following income statement: For the Year Ended December 31, 2010 Sales revenue ......... $2,460,000 Cost of goods sold...
-
1. Prepare a graph of the monthly forecasts and average forecast demand for Chicago Paint Corp., a manufacturer of specialized paint for artists. Compute the demand per day for each month (round your...
-
A 50-MHz generator with Zg = 50 is connected to a load ZL= 50 j25- . The time-average power transferred from the generator into the load is maximum when Zg = Z_L_ where Z _L is the complex...
-
Sigma bonds experience free rotation at room temperature: In contrast, Ï bonds do not experience free rotation. Explain. . cec - `H
-
Consider the reaction below. The rate of this reaction is markedly increased if a small amount of sodium iodide is added to the reaction mixture. The sodium iodide is not consumed by the reaction and...
-
What is always true about a foodservice employees tips? a. They are taxable income. b. They are nontaxable income. c. They are controlled by managers. d. They cannot include any mandatory tip pooling...
-
Blooms Ltd. management recently reported adhering to a target capital structure. The company has the following target: a debt ratio of 50%. The book debt ratio is however 20% on total assets of R1...
-
How has Technology changed the way all industries communicate and maintain documents? What are your experiences and how comfortable are you with Microsoft WORD?
-
1) Please write a program to calculate volume of a cylinder (V = rh). Prompt the user to enter values for radius and height. Use 3.14 for . Only two decimal places. Print out the values in the...
-
How do you determine what an event is "worth"? What resources (local, state, regional, national) are available to event planners to aid with acquiring event funding? What are the social, cultural,...
-
What is the correct entry to record gross loss? Is it debit P&L and credit Trading?
-
On January 10, 2017, a fire destroyed a warehouse owned by NP Company. NPs adjusted basis in the warehouse was $530,000. On March 12, 2017, NP received a $650,000 reimbursement from its insurance...
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
Epoxide?s react with Grignard reagents to yield alcohols propose a mechanism. 1. CHMgBr 2. * CH
-
How would you prepare the following substances from Cyclopentanol? More than one step may be required. (a) Cyclopentanone (b) Cyclopentane (c) 1-Methylcyclopentanol (d) Trans-2-Methylcyclopentanol I...
-
What products would you expect to obtain from reaction of 1-methylcyclo-hexanol with the following reagents? (a) HBr (b) NaH (c) H2SO4 (d) Na2Cr2O7
-
Acaba de contratar a un nuevo cliente que lleva ms de un ao en el negocio y que ha estado utilizando Excel para hacer un seguimiento de su actividad empresarial mensual. Qu 2 funciones de QuickBooks...
-
Barrhaven-brew, a local Ottawa micro-brewery, recently merged with a bottle making plant in New York to complement its Barrhaven brewing facilities. Barrhaven-brew has two divisions: Bottle Making...
-
De qu dos formas puedes crear una plantilla personalizada para un proyecto? Duplicar una plantilla de QuickStart Importar una plantilla Descargar proyectos desde la pestaa de proyectos compartidos...

Study smarter with the SolutionInn App


