Propose a mechanism for the following process: REN heat + N2 + CO2
Question:
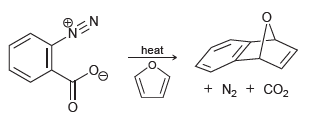
Transcribed Image Text:
REN heat + N2 + CO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
NEN Off R...View the full answer

Answered By

Balram Kumar Pandey
I have been teaching students for more than 5 years, my strongest subject is mathematics. In a span of 2 months I have completed mathematics of 10th class. Those students cleared the board exams.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for the following reaction. H2SO IM
-
Propose a mechanism for the following reaction. excess HBr Br tetrahydrofuran 1,4-dibromobutan
-
Propose a mechanism for the following ring-opening transesterification. Use the mechanism in Problem 21-13 as a model. H+ CH3OH HO OCH,
-
Let n=102 +r, where , re N, 0r9. A number a is chosen at random from the set {1, 2, 3, ..., n} and let pn denote the probability that (a 1) is divisible by 10. 70. If r = 0, then npn equals (a) 22...
-
1. The number of letter pairs that can be formed in any order from any two of the first 13 letters in the alphabet (A-M) is an example of a permutation. 2. The number of permutations of n elements...
-
Take two solid cylindrical specimens of equal diameter but different heights. Assume that both specimens are compressed (frictionless) by the same percent reduction, say 50%. Prove that the final...
-
Find a currency of your choice. How has its value changed against the U.S. dollar over the last ninety days? What about since January 3, 1975?
-
Tucson, a U.S. corporation organized in Year 1, reports the following items for a three-year period. The foreign source and worldwide taxable income items are determined under U.S. law. a. What is...
-
Let h(a) 5-x-3 and let f be the inverse function of h. Notice that h(-1)=7. f'(7)=
-
During the current year, Central Auto Rentals purchased 60 new automobiles at a cost of $24,000 per car. The cars will be sold to a wholesaler at an estimated $8,000 each as soon as they have been...
-
Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the following 1 H NMR and 13 C NMR spectra: 3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4...
-
Phenacetin was widely used as an analgesic before it was removed from the market in 1983 on suspicion of being a carcinogen. It was widely replaced with acetaminophen (Tylenol), which is very similar...
-
Compare and contrast the appearance and content of an ER diagram with that of an REA model.
-
Distinguish between identification and authentication. Is there a relationship between the two? In setting up an authentication scheme, what would be the major factor(s) influencing you to choose...
-
Why should QA personnel not undertake information systems development, implementation, operations, and maintenance work?
-
In the context of boundary-subsystem controls, what is a Trojan-horse threat?
-
Why are arbitrated digital signature schemes sometimes needed?
-
Briefly explain the difference between local PIN validation and interchange PIN validation. Why is a PIN checkdigit useful when interchange PIN validation must be used?
-
McCormick Ltd., a public company following IFRS 16, recorded a right-of-use asset and lease liability at $150,000 on May 1, 2020. The interest rate is 10%. McCormick made the first lease payment of...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Rank the following carbocations in decreasing order of stability. Classify each as primary, secondary, or tertiary. (a) The isopentyl cation, (b) The 3-methyl-2-butyl cation, (c) The 2-methyl-2-butyl...
-
Each of the following proposed mechanisms for the free-radical chlorination of methane is wrong. Explain how the experimental evidence disproves each mechanism. (a) Cl2 + hv Cl2* (an activated form...
-
Rank the following radicals in decreasing order of stability. Classify each as primary, secondary, or tertiary. (a) The isopentyl radical, (b) The 3-methyl-2-butyl radical, (c) The 2-methyl-2-butyl...
-
Hawk Homes, Inc., makes one type of birdhouse that it sells for $29.70 each. Its variable cost is $13.30 per house, and its fixed costs total $14,169.60 per year. Hawk currently has the capacity to...
-
Sales $710,000 Cost of goods sold 182,000 Gross margin 528,000 Selling and administrative expenses 151,900 Net operating income 376,100 Nonoperating items: Gain on sale of equipment 7,000 Income...
-
In class we have been assuming that all of government spending falls on domestic goods. Assume instead that a fraction falls on imports (such as capital equipment and military hardware): mg =...

Study smarter with the SolutionInn App


