Propose a structure for ether with molecular formula C 7 H 8 O that exhibits the following
Question:
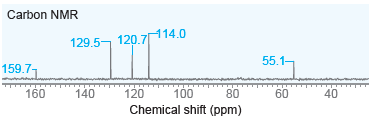
Transcribed Image Text:
Carbon NMR 114.0 129.5, 120,714.0 55.1- 159.71 160 120 140 100 100 Chemical shift (ppm) 80 80 60 60 40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)

Answered By

Farhan Ansari
I Farhan Ansari, I have completed graduation and post graduation from AMU and currently I am doing PhD in statistics.When I started my graduation I also joined coaching as teachers. I love solving mathematics as well as Statistics with concept.I want to anyone challenge to me in my field.I like teaching and tutoring.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Two constitutional isomers having molecular formula C4H6O are both symmetrical in structure. In their infrared spectra, neither isomer when in dilute solution in CCl4 (used because it is nonpolar)...
-
Propose a structure for an alcohol with molecular formula C5H12 O that has the 1H NMR spectrum given in Fig. 9.46. Assign the chemical shifts and splitting patterns to specific aspects of the...
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
An SAT prep course claims to increase student scores by more than 60 points, on average. To test this claim, 9 students who have previously taken the SAT are randomly chosen to take the prep course....
-
Assume that the Hamiltonian cycle problem is NP-complete for undirected graphs. a. Prove that the Hamiltonian cycle problem is NP-complete for directed graphs. b. Prove that the unweighted simple...
-
Explain what is wrong with the statement that in the Monty Hall Three-Door Puzzle the probability that the prize is behind the first door you select and the probability that the prize is behind the...
-
The following is the statement of financial position of TT and Co. (see Self-Assessment Question 3.1 on page 104) at the end of its first year of trading: During 2016, the following transactions took...
-
Police are to conduct random breath testing on drivers on a busy road one Friday evening. Suppose 3% of the drivers drink and drive at the time. Let X be the number of drivers that police need to...
-
Photos-2022-04-28 (18).png 79F Clear Skipped [The following information applies to the questions displayed below.] Laker Company reported the following January purchases and sales data for its only...
-
Larry Power started a new business in the name of Power Electrical on October 1, 2020. During October, a number of activities occurred and the following totals resulted at October 31, 2020 (shown in...
-
The risk-averse person will buy health insurance a. Only if the expected health costs equal the insurance premium. b. Only if the expected health costs are greater than the insurance premium. c. Even...
-
Propose a structure for a compound with molecular formula C 8 H 18 O that exhibits the following 1 H NMR and 13 C NMR spectra. Proton NMR 3.0 2.0 Chemical shift (ppm) 4.0 3.5 2.5 1.5 1.0 0.5 Carbon...
-
Determine whether the function is even, odd, or neither. (a) (x) = x 5 (b) g(t) = t 3 t 2 (c) F(t) = 1/t 4 + t 2
-
Cory and Cassandra's mother/mother-in-law, Margaret, issued a $1 000 000 twenty-five year interest-only loan to the couple. Under the terms of the loan, they make annual payments of interest every...
-
Examine a company where a leader effectively managed a crisis. What leadership traits and strategies did they employ to navigate the crisis and maintain team morale? Explain
-
5 kg/s of steam at 5MPa and 600C is throttled in a valve to 3MPa. Determine the entropy generation rate per unit time.
-
What street used to be located where the Gibbs - Green plaza is location?
-
The world has recently faced crises like the Covid -19 epidemic coupled with the Ukraine War. The impact has been huge and diverse on all of us. Similarly, Life crisis is an inevitable phenomenon for...
-
How is the hatching of water python eggs influenced by the temperature of the snakes nest? Researchers randomly assigned newly laid eggs to one of three water temperatures: hot, neutral, or cold. Hot...
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
The absorption for the hydrogens of benzene appears 444Hz downfield from TMS on an instrument that operates of 60MHz. (a) Calculate the position of this absorption in units. (b) Calculate the...
-
How many absorption are expected in the 1 H-NMR spectra of these compounds? CCH; . ) C,H,CH, c) CH;CH,CHCH3 b) " CH3 C=C - . d) -, e) C= -
-
Predict the approximate chemical shifts for the different hydrogen's in thesecompounds: CI CI a) CH,CH,CH3 b) CH;CHCH3 c) CH,COCH,CH3 d) CH;CHCH2
-
A physical inventory count showed XYZ Company had inventory costing $319,000 on hand at December 31, 2023. This amount did not include the following: 1. Inventory costing $26,000 that was shipped to...
-
Nicholas Company manufacturers TVs. Some of the company's data was misplaced. Use the following information to replace the lost data: Analysis Actual Results Flexible Variances Flexible Budget Sales-...
-
The following information was taken from the financial records of Stormer Company for the year ended December 31, 2023 Retained earnings balance at December 31, 2023 Cash dividends declared for the...

Study smarter with the SolutionInn App


