Propose an efficient synthesis for each of the following compounds using the acetoacetic ester synthesis. (a) (b)
Question:
(a)
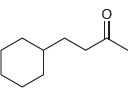
(b)
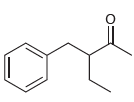
(c)
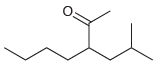
(d)
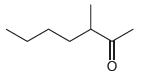
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a b c d ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH H.
-
Propose an efficient synthesis for each of the following transformations: a. b. c. d. Br
-
Describe two methods for determining the molecular mass of a polypeptide. Which is more accurate and why?
-
An independent polling organization interviewed 100 college students to determine their political party affiliations and whether they favor a balanced-budget amendment to the Constitution. The table...
-
Data for McLanie Company are presented in BE6-2. Compute the cost of the ending inventory under the average-cost method. (Round the cost per unit to three decimal places.) In BE6-2 In its first month...
-
Lessee Company enters into a 6-year finance lease of non-specialized equipment with Lessor Company on January 1,2020 . Lessee has agreed to pay \(\$ 28,000\) annually beginning immediately on January...
-
Presented below is information related to Brokaw Corp. for the year 2012. Instructions(a) Prepare a multiple-step income statement for 2012. Assume that 60,000 shares of common stock are...
-
I need help with this one. The Blue Giant has a profit margin of 6.2% and a dividend payout ratio of 40%. The total asset turnover is 0.9259, and the debt-equity ratio is .54. What is the sustainable...
-
This exercise uses the function MapColor and predicates In(x, y), Borders(x, y), and Country(x), whose arguments are geographical regions, along with constant symbols for various regions. In each of...
-
In Problem 22.38, we saw an intramolecular example of a malonic ester synthesis using excess base and 1,4-dibromobutane. If this dibromide is used in an acetoacetic ester synthesis, an intramolecular...
-
Guanidine is a neutral compound but is an extremely powerful base. In fact, it is almost as strong a base as a hydroxide ion. Identify which nitrogen atom in guanidine is so basic, and explain why...
-
In combining operators sequentially, it is useful to insert an arbitrary function after the operator to avoid errors. For example, if the operators A and B are x and d /dx, then AB f (x) = x df...
-
Describe the marketing channels you will use to promote your t-shirt company to reach your targeted market? Describe the benefit and goal for each channel? Describe how you will measure the Return on...
-
The pacesetting leadership style is primarily focused on achieving the end goal, driving high-performing team members to perform at their highest possible rate. This emotionally-driven leadership...
-
How were these goals passed down to the managers or adapted to fit the manager's team in United Health Group? Did it align to the business strategies? How did the manager's leadership style affect...
-
Liz purchased two acres of lakefront property from her ex-husband, Richard, paying $500,000. Richard conveyed the property via a quitclaim deed. Liz's attorney dutifully recorded the deed the next...
-
Complete these equations by predicting the major product formed in each reaction. Note that some of these reactions involve rearrangements. (See Examples 5.2, 5.5) (a) HBr (b) HSO + HO (c) HSO + HO...
-
Repeat Exercise 2 for the statistics quiz score that was part of Ihno's experiment. Data from exercise 2 Use your statistical package to create a z-score variable corresponding to the math background...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
In each pair of compounds, which compound has the higher boiling point? Explain your reasoning. (a) octane or 2,2,3-trimethylpentane (b) Nonane or 2-methylheptane (c) 2, 2, 5-trimethylhexane or nonane
-
Analyze the balance sheet for LMN Co. as of December 31, 2023, to evaluate its operational efficiency and profitability. LMN Co. Balance Sheet As of December 31, 2023 Assets Current Assets Cash...
-
The pension plan assets balance is reported Multiple choice question. in the disclosure notes. in the statement of cash flows. in the balance sheet. in the income statement
-
True or False? General ledger accounts help ensure that all expenses and revenues are allocated to the correct department. Each department in the organization will have its own department codes that...

Study smarter with the SolutionInn App


