Starting with sodium azide as your source of nitrogen and using any other reagents of your choice,
Question:
Compounds in 23.18
(a)
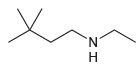
(b)
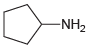
(c)
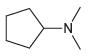
(d)
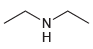
(e)
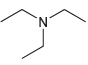
(f)
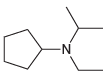
Transcribed Image Text:
-NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The first alkyl group ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using ammonia as your source of nitrogen, show the reagents you would use to prepare each of the following amines: (a) (b) (c) (d) (e) (f) Z
-
Starting with the following compound and using any other reagents of your choice, outline a synthesis for trimyristin. H.
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
Rice and Flower were partners sharing profit and loss equally. Statement of Financial Position as at 31 December 2020. Non current Assets Premises Machinery Vehicles Fittings Current Assets Inventory...
-
Using the following data for a hypothetical economy, calculate gross domestic product (GDP), net domestic income (NDI), and government budget surplus (BS):...
-
Chemically modified steroids have become increasingly important in medicine. Give the possible product(s) of the following reactions. In each case, identify the major stereoisomer formed on the basis...
-
The budget for the University Printing Company for 2007 follows. The company typically uses a so-called cost-plus pricing system. Direct material and direct labour are computed, overhead is added at...
-
The following transactions were incurred by Whooley Fabricators during January, the first month of its fiscal year. Requirements 1. Record the proper journal entry for each transaction. a. $ 205,000...
-
7. Let M(t) represent the rate at which people enter Macy's department store, in people per hour, t hours into their Black Friday sale. Interpret the statement M(3.5) = 432 in the context of this...
-
The following questions refer to the tree of Figure 8.3.a. Which node is the root?b. What are the internal nodes?c. How many descendants does node cs016/ have?d. How many ancestors does node cs016/...
-
Draw the structure of each of the following compounds: (a) Cyclohexylmethylamine (b) Tricyclobutylamine (c) 2,4-Diethylaniline (d) (1R,2S)-2-Methylcyclohexanamine (e) ortho-Aminobenzaldehyde
-
What is the difference between a firms mission and its objectives? Why is it important that both are conveyed clearly to employees and to customers?
-
Draw the graph of the function and state the domain and range. y = (1/4) x
-
What is the logic of designing echeloned logistical structures? Can echeloned and direct structures be combined?
-
Many investors rely on highlights from the annual report as provided by online and CD-ROM-based information services and do not read the reports themselves. These services do not contain the text of...
-
Discuss the major supply chain APS applications with particular focus on the role and anticipated benefits for each application.
-
How could the relationship between the auditor and management change if conclusive proof emerged that a qualified audit report had adverse implications?
-
Discuss how error accountability can be a major factor in improving forecast performance.
-
Assuming you are an agent with the Federal Bureau of Investigation, do the following: I. Plan and coordinate an investigation in a manner that would not arouse suspicion fro m Cummings and Baptiste....
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
An all-suprafacial [3,3] sigma tropic rearrangement could in principle take place through either a chair-like or a boat-like transition statel When the terpene germacrone is distilled under reduced...
-
Explainusing (a) resonance argumeots and (b) molecular orbital arguments why the unpaired electron in the allyl radical is delocalized to carbon-l and carbon-3 but not to carbon-2.
-
Explainusing (a) resonance argumeots and (b) molecular orbital arguments why the unpaired electron in the allyl radical is delocalized to carbon-l and carbon-3 but not to carbon-2.
-
Japan Company produces lamps that require 2 standard hours per unit at a standard hourly rate of $18.30 per hour. Production of 7,900 units required 15,330 hours an hourly rate of $18.70 per hour....
-
Investors may accept an asset with greater risk, only if the asset. Multiple Choice costs less costs more offers greater risk premium offers less return
-
Paver Corporation is making a $102,000 investment in equipment with a 5-year life. The company uses the straight-line method of depreciation and has a tax rate of 20 percent. The companys required...

Study smarter with the SolutionInn App


