Tetrahydrofuran (THF) can be formed by treating 1, 4-butanediol with sulfuric acid. Propose a mechanism for this
Question:
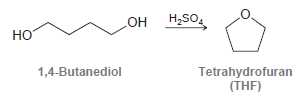
Transcribed Image Text:
H,SO, H;SO. Он но 1,4-Butanediol Tetrahydrofuran (THF)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
H O...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for the following transformation: [H,SO,] Meo
-
Propose a mechanism for the following transformation: . 1) Excess LA, 2) H20
-
Local 54 has retained James Love to represent it in a grievance against Dilated Peoples Optical Inc. The union is grieving the employers decision to exclude from the bargaining unit the position of...
-
Parlee Company's sales are 30% in cash and 70% on credit. Sixty % of the credit sales are collected in the month of sale, 25% in the month following sale, and 12% in the second month following sale....
-
Exhaust gas at 400oC and 1 bar from internal-combustion engines flows at the rate of 125 mol s-1 into a waste-heat boiler where saturated steam is generated at a pressure of 1,200 kPa water enters...
-
Location of retail outlets is an issue in strategic planning. What initial steps would you recommend to Juanita (see Application Question 2) when she considers a location for her store?
-
Zocco Corporation has an inventory conversion period of 75 days, an average collection period of 38 days, and a payables deferral period of 30 days. a. What is the length of the cash conversion cycle...
-
P22-48B Preparing an operating budget-sales, production, direct materials, direct labor, overhead, COGS, and S&A expense budgets The Haney Batting Company manufactures wood baseball bats. Haney's two...
-
Start with the partial model in the file Ch06 P15 Build a Model.xlsx on the textbook's Web site. The file contains hypothetical data for working this problem. Goodman Corporation's and Landry...
-
When 1, 4-dioxane is heated in the presence of HI, compound A is obtained: a. Draw the structure of compound A. b. If one mole of dioxane is used, how many moles of compound A are formed? c. Show a...
-
When ethylene glycol is treated with sulfuric acid, 1, 4-dioxane is obtained. Propose a mechanism for this transformation: H,SO, Ethylene glycol 1,4-Dioxane
-
In 2015, HD had reported a deferred tax asset of $98 million with no valuation allowance. At December 31, 2016, the account balances of HD Services showed a deferred tax asset of $130 million before...
-
Based on the data above, calculate the price elasticity of supply coefficient of wine between January 2010 and January 2011. From the answer choices below, SELECT TWO answers. 1. Select one number...
-
Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.35 M acetic acid + 0.21 M sodium acetate 0.31 M ammonium bromide + 0.30 M ammonia 0.18 M calcium hydroxide...
-
Two boxes, A and B, are stacked on top of each other on top of a conveyor belt as shown below. The values of the coefficients of friction between the two boxes are k = 0.1 and =0.2. The values of the...
-
What would be the total appraisal cost appearing on the quality cost report? What would be the total internal failure cost appearing on the quality cost report? What would be the total external...
-
Question 1 Study the scenario and complete the question(s) that follow: 20 Marks Setting Up Oracle SQL Developer is Oracle's free application for accessing and working with Oracle databases. This...
-
What is the interpretation of a probability value of a. .00? b. .20? c. .50? d. .75? e. 1.00?
-
Medi-Exam Health Services, Inc. (MEHS), located in a major metropolitan area, provides annual physical screening examinations, including a routine physical, EKG, and blood and urine tests. MEUS's...
-
What is the structure of the a-keto acid formed by transamination of each of the following amino acids? (a) Threonine (b) Phenylalanine (c) Asparagine
-
What enzyme cofactor is associated with each of the following kinds of reactions? (a) Transamination (b) Carboxylation of a ketone (c) Carboxylation of an -keto acid
-
The glycolysis pathway shown in figure has a number of intermediates that contain phosphate groups. Why can 3-phosphoglyceryl phosphate and phosphoenolpyruvate transfer a phosphate group to ADP while...
-
Yolanda tiene una tienda de productos de computacin, su objetivo principal es poder mantener econmicamente a su familia, pues la venta de computadoras es un buen negocio. La situacin mostrada de...
-
The income statement of Novak Co. for the month of July shows net income of 3,280 based on Service Revenue 7,750, Salaries and Wages Expense 2,920, supplies expense 930, and Utilities Expenses 620....
-
Soru -2- Aadaki ilemleri banka ve merkez bankas bilanolarnda t tablolar ile gsteriniz. 1 - Mteri A Bankasndaki senetleriyle bir araba satn alyor. 2 - A Bankas B Bankasndan FED Fonlarn bor alyor. ki...

Study smarter with the SolutionInn App


