The following mass spectrum is for octane. a) Which peak represents the molecular ion? b) Which peak
Question:
a) Which peak represents the molecular ion?
b) Which peak is the base peak?
c) Draw the structure of the fragment that produces the base peak.
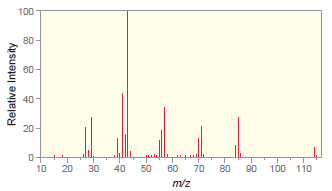
Transcribed Image Text:
100 80 60 40 20 110 10 20 30 40 50 60 70 80 90 100 m/z Relative Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
a The molecular io...View the full answer

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2-Methylpentanc (C 6 H 14 ) has the mass spectrum shown. Which peak represents M + ? Which is the base peak? Propose structures for fragment ions of m/z = 71, 57, 43, and 29. Why does the base peak...
-
Calculate the mass of octane, C8H18(l), that must be burned in air to evolve the same quantity of energy as produced by the fusion of 1.0 g of hydrogen in the following fusion reaction: Assume that...
-
Use Figure 24-15 to suggest which type of liquid chromatography you could use to separate compounds in each of the following categories: (a) Molecular mass < 2 000, soluble in octane (b) Molecular...
-
In Exercises 7980, find the value of y if the line through the two given points is to have the indicated slope. (3, y) and (1, 4), m = -3
-
Write an essay about a "Magazine" focusing on the demographics, describing the niches the magazine is trying to reach. For this the chosen magazine is: Elle magazine. Some demographics that should be...
-
Let S22 be the vector space of 2 Ã 2 symmetric matrices. Find a basis for S22 composed of eigenvectors of the linear transformation Q: S22 S22. 25a 18b30c -16a 11b 20e 16a 11b-20c -1a 9b 12c
-
What sort of a discount can a minority shareholder get compared with a financial value? Show how the situation differs between a listed company and an unlisted company.
-
Sundance Skis Company is preparing its end-of-year gross margin computations. Sundance Skis manufactures three types of products: skis, snowboards, and snow skates. The following information, as of...
-
Write an equation for a Vertical asymptotes at x intercepts at x = y intercept at 8 y =
-
The bond indenture for the 10-year, 9% debenture bonds issued January 2, 2013, required working capital of $100,000, a current ratio of 1.5, and a quick ratio of 1.0 at the end of each calendar year...
-
Propose a molecular formula that fits the following data. a) A hydrocarbon (C x H y ) with a molecular ion peak at m/z = 66 b) A compound that absorbs IR radiation at 1720 cm -1 and exhibits a...
-
Calculate the HDI for each molecular formula. a) C 4 H 6 b) C 5 H 8 c) C 40 H 78 d) C 72 H 74 e) C 6 H 6 O 2 f) C 7 H 9 NO 2 g) C 8 H 10 N 2 O h) C 5 H 7 Cl 3 i) C 6 H 5 Br j) C 6 H 12 O 6
-
1. Find the energy of a photon of electromagnetic radiation with frequency 9.20 x 1016 Hz. 2. Find the energy of a red photon. 3. Find the energy of a blue photon. 4. Find the energy of a yellow...
-
For your organization and incident investigations, how can you use Dr. Reason's Swiss Cheese model in the development of your company's or your future company's incident investigation program? Is...
-
A professor is a member of a professional organization and is mostly interested in the newsletters sent and in participating in conferences. What benefit is the professor primarily seeking?Explain
-
When I invested $50,000 in this mutual funds at the beginning of the 3rd year, how much the invested money would be worth at the end of the year 8??State each step of calculation and explain the...
-
How can an organization ensure that they are asking the right questions during the market research process?
-
Describe what actions an organization can take to encourage employees to think more creatively. Describe the characteristics of an effective control system.
-
Suppose we are given two sequences A and B of n integers, possibly containing duplicates, in the range from 1 to 2n. Describe a linear-time algorithm for determining if A and B contain the same set...
-
The outer loop controls the number of students. Note that the inner loop of this program is always executed exactly three times, once for each day of the long weekend. Modify the code so that the...
-
Is there an unbranched alkane containing 23 hydrogen atoms? If so, give its structural formula; if not, explain why not.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
DUX COMPANY Comparative Balance Sheets December 31, 2018 and 2017 ($ in 000s) 2018 2017 Assets Cash Accounts receivable Less: Allowance for uncollectible accounts $ 39 $ 23 47 53 (5) (4) Dividends...
-
Frank Incorporated is trying to decide whether to lease or purchase a plece of equipment needed for the next 10 years. The equipment would cost $53,000 to purchase, and maintenance costs would be...
-
Skull Company makes snowboards and uses the total cost method in setting product price. Its costs for producing 12,500 units follow. The company targets a 10.0% markup on total cost. Variable Costs...

Study smarter with the SolutionInn App


