The following triene reacts with excess maleic anhydride to produce a compound with molecular formula C 14
Question:
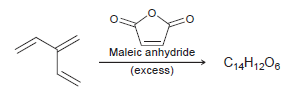
Transcribed Image Text:
Maleic anhydride (excess) C14H1208
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider a process that attempts to prepare tyrosine using a HellVolhardZelinski reaction: (a) Identify the necessary starting carboxylic acid. (b) When treated with Br 2 , the starting carboxylic...
-
Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C 10 H 18 , on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the...
-
A mixture of maleic anhydride and benzoic acid containing 10 mol% acid is a product of the manufacture of phthalic anhydride. The mixture is to be distilled continuously in a column with a total...
-
On September 1, 2025, Swifty Corporation acquired Windsor Enterprises for a cash payment of $800,000. At the time of purchase, Windsor's balance sheet showed assets of $570,000, liabilities of...
-
The state of plane stress in a body is described by the following stresses: 1 = 8500 kN/m2 compression, 3 = 1500 kN/m2 tension. Determine by means of the Mohr circle the normal stress and shear...
-
Suppose yn represents the population of the world the nth year after 1 800. That is. y0 is the population in 1800, y1 is the population in 1801 and so on. From the definition of the derivative as the...
-
What are two numbers with an absolute value 87 ?
-
The following information is available for two different types of businesses for the 2016 accounting year. Hopkins CPAs is a service business that provides accounting services to small businesses....
-
Avery Corporation's target capital structure is 35% debt, 10% preferred, and 55% common equity. The interest rate on new debt is 6.50%, the yield on the preferred is 6.00%, the cost of common from...
-
How would you measure the level of customer satisfaction with your favorite restaurant?
-
Government inspectors who check on the quality of services provided by retailers as well as government requirements for licensing in various professions are both attempts to resolve: a. The moral...
-
True or False: A market may collapse and have relatively few transactions between buyers and sellers if buyers have more information than sellers.
-
Suppose colors as indicated below represent numerals in a base 4 numeration system. Write the HinduArabic numerals equivalent to each of the following. = 3
-
Why is it not a good idea to delay payroll tax payments to the government?
-
Discussing audit findings with an auditee in advance of drafting a report is most advantageous when: a. There is agreement on the fact. b. Fraud is suspected. c. Corrective action is needed. d. The...
-
What causes the disparity between the initial offering price and the trading price afterward?
-
How will you measure your success in your business or entrepreneurial career?
-
After internal auditors report deficiency findings that have not yet been corrected, they should: a. Correct the defect themselves. b. Dismiss the matter from further consideration. c. Evaluate the...
-
Write a Java program for a matrix class that can add and multiply arbitrary twodimensional arrays of integers.
-
Which of the following raises the credibility of areport? Which of the following raises the credibility of a report? Multiple Choice avoiding predictions avoiding the use of cause-effect statements...
-
Why do you suppose ketone halogenations in acidic media referred to as being acid-catalyzed, whereas halogenations in basic media are base-promoted? In other words, why is a full equivalent of base...
-
How could you use a malonic ester synthesis to prepare the following compounds? Show allsteps. (b) CH3CH2CH2CHC CH (c) CHCH2C CHCHCH-CH,C C
-
Monoalkylated and dialkylated acetic acids can be prepared by the malonic ester synthesis, but trialkylated acetic acids (R3CCO2H) cant be prepared. Explain
-
is it true that In the 1 9 8 0 s , the government trend toward deregulation made it easier for corporations to take over or merge with companies in different sectors of the economy or companies in...
-
If both the demand and supply of financial capital were to rise, we would expect Question 2 0 options: a ) the interest rate to definitely rise b ) the interest rate to definitely fall c ) the...
-
The National Institute of Standards and Technology of the U.S. Department of Commerce has a special publications series on IT security. The webpage listing these 800 series publication...

Study smarter with the SolutionInn App


