When the following ester is treated with lithium iodide in DMF, a carboxylate ion is obtained: (a)
Question:
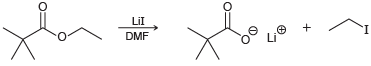
(a) Draw the mechanism of this reaction.
(b) When the methyl ester is used as the substrate, the reaction is 10 times faster:
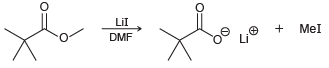
Explain the increase in rate.
Transcribed Image Text:
LiI DMF LiI Mel DMF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a b This reaction occurs via an SN2 process As s...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An ether extraction of nutmeg gives large quantities of trimyristin, a waxy crystalline solid of melting point 57 C. The IR spectrum of trimyristin shows a very strong absorption at 1733 cm-1. Basic...
-
The compound below is an example of a methyl ester. Methyl esters react with lithium iodide to give lithium carboxylate salts. The solvent in this example is pyridine (margin). Suggest several...
-
When the following compound is treated with excess methyl iodide, a quaternary ammonium salt is obtained that bears only one positive charge. Draw the structure of the quaternary ammonium salt. *NH2
-
The lease agreement and related facts indicate the following: a. Leased equipment had a retail cash selling price of $300,000. Its useful life was five years with no residual value. b. The lease term...
-
How does a "line of credit" work? What type of business would find having one particularly desirable?
-
The following compounds, listed with their boiling points, are liquid at -10C: butane, -0.5C; ethanol, 78.3C; toluene, 110.6C. At -10C, which of these liquids would you expect to have the highest...
-
What is the link between motivation, performance, and rewards?
-
Provide a short explanation of the difference between each of the following sets of terms: a. Bound control versus unbound control b. Design mode versus run mode c. Symbol versus symbol on a forms...
-
ICS Finance Corp. borrowed money by issuing $1,000,000 of 6 percent bonds payable at 98.5. a. How much cash did ICS receive when it issued the bonds payable? b. How much must ICS pay back at...
-
Allison Hopkins has her own chain of music stores, Hopkins Music. Her stores sell musical instruments, sheet music, and other related items. Music lessons and instrument repair are also offered...
-
Identify the reagents that you would use to accomplish each of the following transformations: a. b. c. d. e. f. -Br OH
-
It would cost the town of Irondale $50 million to build a gas-powered generator that could produce a maximum of 5 megawatts of electricity at 15 cents per hour. Another alternative would be for...
-
The graph of a linear function f is shown. (a) Identify the slope, y-intercept, and x-intercept. (b) Write a formula for f. (c) Estimate the zero of f. -3-2-1 3 23
-
Consider the curve f ( x ) = ( - 1 / 3 x - 1 ) square root ( - 2 x - 9 ) and the line g ( x ) = - x + 7 Find the equation of a tangent line to f ( x ) that is parallel to g ( x ) . Give your equation...
-
Reggie has just taken over management of a family business. He wants to make sure that it makes financial sense to keep the business going. He could sell the building today for $480,000. Keeping the...
-
Assume the following information, i. Mexican one-year interest rate = 15 percent ii. U.S. one-year interest rate = 11 percent iii. If interest rate parity exists, what would be the forward premium or...
-
Draw the logic circuit for the following Boolean equation. Simplify the equation and construct the truth table of the circuit using the simplified equation. F= (A + B). (C+A) + BC +A
-
As part of a quality-assurance program being initiated by Community Hospital, each vice president personally interviewed every staff member under his or her jurisdiction to determine what actions...
-
A group of researchers estimates the mean length of time (in minutes) the average U.S. adult spends watching television using digital video recorders (DVRs) and other forms of time-shifted television...
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
Disparlure, C19H38O, is a sex attractant released by the female gypsy moth, Lymantria dispar. The 1H NMR spectrum of disparlure shows a large absorption in the alkane region, 1 to 2 , and a triplet...
-
How would you synthesize racemic disparlure (Problem 18.46) from corn- pounds having ten or fewer carbons?
-
Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction. Ph H30+ PHCHCH Ph Ph
-
The transactions in the journal and the accounts in the ledger are linked together by a transfer process called:Group of answer choicesaudit trailmigratingpostingbalancing
-
Garbo township recorded it's police departments appropriations for the year ending June 30, 2025. Police appropriations had increased to $7,600,000. During the year's first budgetary month ending...
-
QUALITY INDICATOR: Ninety-five percent of physician appointments/reappointments will be completed within 90 days of receipt of all required materials. medical staff office health information business...

Study smarter with the SolutionInn App


