When the following optically active alcohol is treated with HBr, a racemic mixture of alkyl bromides is
Question:
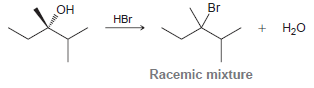
Draw the mechanism of the reaction, and explain the stereochemical outcome.
Transcribed Image Text:
Br Он HBr + H20 Racemic mixture Мн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
The chirality center is lost when the leaving g...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When 2-phenyl-2-butanol is allowed to stand in ethanol containing a few drops of sulfuric acid, the following ether is formed: Suggest a reasonable mechanism for this reaction based on the...
-
Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B. In a separate reaction, A is treated with PBr3, converting A into compound C. Compound C is...
-
When a primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out...
-
You're pretty sure that your candidate for class president has about 55% of the votes in the entire school. But you're worried that only 100 students will show up to vote. How often will the underdog...
-
What is WBGT?
-
Name some industries in which job order costing is common.
-
Long-Term Debt divided by population ratio is decreasing over time. m. Current Liabilities divided by Gross Revenues ratio is increasing over time. n. Tax Levy divided by Tax Limit ratio is...
-
Harry's Hardware does a brisk business during the year. During Christmas, Harry's Hardware sells Christmas trees for a substantial profit. Unfortunately, any trees not sold at the end of the season...
-
Mark has recently begun working at a local fruit and vegetable grocer. Mark spends a good deal of his time talking to customers and fulfilling sales orders. Over time, he identifies a weakness in ...
-
Aubrae and Tylor Williamson began operations of their furniture repair shop (Furniture Refinishers, Inc.) on January 1, 2019. The annual reporting period ends December 31. The trial balance on...
-
What kind of food did the extinct equid likely eat?
-
When ethylbenzene is treated with NBS and irradiated with UV light, two stereoisomeric compounds are obtained in equal amounts. Draw the products and explain why they are obtained in equal amounts....
-
The state of stress at a point is given by: \[ \sigma_{x}=60 M P a, \sigma_{\mathrm{y}}=-30 M P a, \tau_{\mathrm{xy}}=30 M p a(c c w) \] Calculate (a) principal stresses, (b) principal planes, (c)...
-
What does it mean to have an emotional understanding of other people?
-
What would be the appropriate operating system architecture for the: (a) Inertial measurement system (b) Nuclear monitoring system (c) Patient monitoring system (d) Airline reservations system (e)...
-
What functions are performed in effective groups? Illustrate how a breakdown in one of these functions can jeopardize the success of the other functions. As your example for class discussion, use the...
-
Show with an example that EDF is no longer an optimal scheduling policy if preemption is not allowed.
-
Outline the general characteristics of a group.
-
Go to https://tinyurl.com/slaterca14e to access Amazons 2016 Annual Report. Go to page 37 and find the balance of interest income in 2016.
-
Determine by direct integration the values of x for the two volumes obtained by passing a vertical cutting plane through the given shape of Fig. 5.21. The cutting plane is parallel to the base of the...
-
Predict the geometry at the carbon of these compounds: a) H-C=N: b) 0: H-C-0-H c) H T CI-C-H T CI
-
Predict the geometry of the following compounds at the indicated atoms: a) H H-C-0-H H At the carbon At the oxygen b) H. H C-N-H At the carbon At the nitrogen c) H. C=O-H H At the carbon At the oxygen
-
Predict the direction of the dipole moments of these compounds: a) H H b) F H-C-H H H H-C-C=N H
-
A manufacturer of microwave ovens exports its products to another country at a price lower than the cost of production in the home country in order to quickly gain market share. This practice known...
-
[ads] Meghan is creating an ad campaign targeting college students. She is deciding between two school newspapers to display her ad. She wants to use the most efficient media vehicle. The Alligator...
-
Question 29 As per our discussion in Lecture, the total percentage of exports that Canada trades with the United States is? Question 29 options: 65% 70% 76% 78% 80% Question 30 A key issue in trade...

Study smarter with the SolutionInn App


